
Gilead First
How a Monopoly on Remdesivir Led to Rationing
What happens when one corporation is given a monopoly on a potentially useful treatment during a pandemic? This report traces the case of remdesivir, an unapproved investigational drug for COVID-19 developed by Gilead Sciences and the U.S. government. Remdesivir is in shortage. The treatment has received emergency use authorization in several countries for COVID-19 patients with severe disease.[1] While there remains uncertainty about the extent of its effectiveness, remdesivir is being rationed.
In Pennsylvania, patients are entering lotteries for a chance to receive the drug.[2] In Florida, politicians are publicly pleading with the federal government for more courses.[3] Across the country, doctors are making tough decisions about who gets access. “Every morning I wake up with palpitations because I don’t know if we’re going to have enough,” says one physician.[4] Globally, the scarcity is even more acute.
This report documents gaps in supply in the U.S. and around the world.
Since July, we find that at least 38 hospitals in 32 cities in 12 states have reported remdesivir shortages in the U.S.[5] The Trump Administration has not publicly considered purchasing the drug from suppliers other than Gilead.[6] The Trump Administration has also persistently mismanaged distribution, with incompetence or malfeasance further fueling scarcity among states. For example, we find that over a month the federal government distributed 1709 courses more of remdesivir to Ohio than Georgia—even though Georgia had nearly three times as many people hospitalized with COVID-19, and more than twice as many new COVID-19 hospitalizations.[7]
We also estimate potential global need. In addition to the U.S., Gilead has entered into a deal with the European Union for nearly all of its supply through September. The corporation has also licensed some generic producers to supplement its capacity and supply some low and middle-income countries.[8] But much of the world remains excluded. We document Gilead’s current supply, the countries excluded from the license and, where data is available, estimate the number of hospitalized patients with severe COVID-19. From August through September, we estimate Gilead will likely have only 15,300 additional treatment courses available for countries beyond the E.U. and the U.S.[9] In contrast, by the end of September, we estimate there will have been hundreds of thousands of hospitalized patients potentially eligible for treatment with remdesivir in countries excluded from Gilead’s voluntary license and deals. [10] Gilead itself has acknowledged in its latest financial disclosures that “. . . there is no assurance that we will be able to meet global supply needs for remdesivir.”[11]
These national and global shortages are rooted in part in patent monopolies. Remdesivir may or may not prove clinically important in the fight against COVID-19, but this current shortage is a reminder that no single corporation can or should supply the world with a COVID-19 treatment or vaccine. In addition to increasing transparency around distribution, governments around the world should permit low-cost generic suppliers of remdesivir to scale-up global production. It is time to stop putting Gilead first.
Background
Remdesivir is an investigational drug developed by Gilead Sciences and the U.S. government. As we described in The Real Story of Remdesivir, the federal government has played a critical role at every stage of its development, pouring in at least $70 million in taxpayer funding.[12] Given the instrumental role of federal scientists, the federal government likely should be named as a co-owner on key patents.[13]
The U.S. government has not leveraged this to demand lower prices. Gilead, instead, is charging developed countries between $390 per vial to $520 per vial—or $2340 per patient to $3120 per patient.[14] The latter is the price for most payers in the U.S. Researchers at the University of Liverpool estimate that the cost of manufacturing remdesivir at scale with a reasonable profit is $0.93 per dose.[15]
Remdesivir is not a silver bullet for COVID-19.[16] In one randomized trial, the National Institutes of Health found that remdesivir reduced time to clinical recovery compared to a placebo (median recovery time of 11 days v 15 days).[17] That same trial did not reveal a significant reduction in deaths. More data is thus needed to establish whether remdesivir provides clinically meaningful benefit and has a favorable benefit-risk profile for the treatment of COVID-19.
But remdesivir could be helpful. With governments around the world straining under the pandemic, remdesivir could relieve some pressure on hospitals. The effect could be particularly pronounced in low-and-middle- income countries with limited health care capacity.
Tracking Supply
On June 29th, Secretary Azar announced an agreement to purchase remdesivir. The press release provided that:
HHS has secured more than 500,000 treatment courses of the drug for American hospitals through September. This represents 100 percent of Gilead’s projected production for July (94,200 treatment courses), 90 percent of production in August (174,900 treatment courses), and 90 percent of production in September (232,800 treatment courses).[18]
On July 29th, the European Union announced an agreement for 30,000 courses to cover the next few months. It said it was still in negotiations for additional supplies “from October onwards.”[19]
Table 1: Public Remdesivir Purchases from Gilead
| Month | U.S. Treatment Courses | European Treatment Courses | Estimated Total Production |
| July | 94,200 | N/A | 94,200 |
| August | 174,900 | 30,000 | 194,333 |
| September | 232,800 | 258,667 | |
| Total | 501,900 | 30,000 | 547,200 |
Based on these figures, we estimate 15,300 treatment courses may be available until end of September from Gilead for remaining global demand.[20] Gilead has since announced it “expect[s] enough supply to meet global real-time need by beginning of October.”[21] But it has not provided details on its future production capacity or how it defines global need. For example, it is not immediately clear whether Gilead is building surplus capacity to meet demand in the event of a serious increase in COVID-19 cases during the winter, or if instead it expects to narrowly meet currently projected demand. Instead, Gilead has vaguely said that it expects to work with partners to produce over 2 million treatment courses cumulatively in 2020—a figure that includes past donations (240,000 courses) and anticipated production through September (547,200).[22] Pfizer and Hikma Pharmaceuticals recently announced that they would help Gilead shore up supply.[23]
In its latest financial disclosures, Gilead acknowledged that “. . . there is no assurance that we will be able to meet global supply needs for remdesivir.”[24]
Domestic Shortages
The U.S. was the first country to authorize the emergency use of remdesivir. On May 1st, the Food and Drug Administration (FDA) issued an emergency use authorization for remdesivir for COVID-19 patients hospitalized with severe disease. Severe disease includes “patients with low blood oxygen levels or needing oxygen therapy or more intensive breathing support such as a mechanical ventilator.”[25] Initially, the National Institutes of Health issued treatment guidelines mirroring the scope of this authorization. However, in response to limited supplies, the National Institutes of Health recently narrowed its guidance. The guidance now reads:
Because remdesivir supplies are limited, the Panel recommends that remdesivir be prioritized for use in hospitalized patients with COVID-19 who require supplemental oxygen but who are not on high-flow oxygen, noninvasive ventilation, mechanical ventilation, or ECMO (BI).[26]
(On August 28, the FDA expanded the emergency use authorization for remdesivir to any hospitalized patient, irrespective of the severity of disease. The available clinical evidence does not support this expansion.)
HHS began allocating commercial remdesivir on July 6th. To estimate shortages, we compiled all news articles referring to hospital shortages of remdesivir in the U.S. from that date. The FDA defines shortages as “a period of time when the demand or projected demand for the drug within the United States exceeds the supply of the drug.”[27] At the hospital level, we operationalized this by including hospitals that reported a denial of their request for remdesivir from public health agencies, and physicians that reported rationing treatment.
We searched for the terms “remdesivir” and “shortage” in Google News, between July 6th and August 8th. We documented the hospital, city, and state where the shortages were reported. We also performed additional searches for remdesivir supply issues in the ten states with the highest burden of COVID-19 cases.
At least 38 hospitals in 32 cities in 12 states reported remdesivir shortages since July 6th.
In Virginia, the head of a hospital said they are in “essence triaging the use of Remdesivir [sic].”[28] In Florida, one county official pled with the Governor for more remdesivir, noting that the local hospitals are in “desperate need.”[29] In Nevada, the head of the pharmacy board says the State of Nevada has requested “thousands and thousands” of cases of remdesivir, but expected to get only 19 in the next shipment.[30] In South Carolina, one physician described his experience running out of remdesivir:
We have already used our two-week allotment . . . So I have to go now from Wednesday until next Monday before we’ll have any more Remdesivir [sic]. And that becomes critical when you have people who need it... the earlier they get it the better they do.[31]
Table 2: Hospitals Reporting Shortages
| State | City | Health Provider | Article Date | Source |
| AL | Anniston | Regional Medical Center | 7/30/2020 | Aniston Star |
| AZ | Phoenix | HonorHealth | 7/20/2020 | KTAR News |
| CA | San Francisco | Zuckerberg San Francisco General Hospital | 7/22/2020 | KQED |
| CA | Rancho Mirage | Eisenhower Health | 7/21/2020 | KESQ News Channel 3 |
| CA | Salinas | Salinas Valley Memorial Healthcare System | 7/9/2020 | Monterey County Weekly |
| FL | Tarpon Springs | AdventHealth | 7/27/2020 | Tampa Bay Times |
| FL | Clearwater | BayCare | 7/27/2020 | Tampa Bay Times |
| FL | St. Petersburg | HCA | 7/27/2020 | Tampa Bay Times |
| FL | Miami | Baptist Hospital (Baptist Health)[32] | 7/17/2020 | Washington Post |
| FL | Boynton Beach | Bethesda Hospital East (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Boynton Beach | Bethesda Hospital West (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Coral Gables | Doctors Hospital (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Marathon | Fishermen’s Community Hospital (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Homestead | Homestead Hospital (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Tavernier | Mariners Hospital (Baptist Health) | 7/17/2020 | Washington Post |
| FL | South Miami | South Miami Hospital (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Miami | West Kendall Baptist Hospital (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Miami | Baptist Children’s Hospital (Baptist Health) | 7/17/2020 | Washington Post |
| FL | Boca Raton | Boca Raton Regional Hospital (Baptist Health) | 7/23/2020 | Boca Magazine |
| FL | Miami | Jackson Health System | 7/17/2020 | NBC Miami |
| FL | Tampa | Unspecified[33] | 7/26/2020 | YourSun.com |
| FL | Orlando | Unspecified | 7/26/2020 | YourSun.com |
| FL | Jacksonville | Unspecified | 7/26/2020 | YourSun.com |
| FL | Sarasota | Sarasota Memorial Health Care System | 7/21/2020 | YourSun North Port |
| FL | Fort Meyers | Lee Health | 7/8/2020 | Fort Meyers News-Press |
| GA | Gainesville | Northeast Georgia Medical Center | 7/10/2020 | The Gainesville Times |
| LA | Baton Rouge | Our Lady of the Lake Regional Medical Center | 8/4/2020 | The Advocate |
| MO | Springfield | Cox Health | 7/13/2020 | Springfield News-Leader |
| MO | Springfield | Mercy Hospital | 7/14/2020 | Springfield News-Leader |
| NV | Las Vegas | St. Rose Dominican | 8/5/2020 | Channel 8 News Now Las Vegas |
| PA | Pittsburgh | University of Pittsburgh | 7/23/2020 | The New York Times |
| SC | Kingstree | Williamsburg Regional Hospital | 7/31/2020 | WCSC |
| SC | Columbia | Prisma Health Richland Hospital | 7/14/2020 | WIS News 10 |
| TX | Houston | Houston Methodist Hospital | 7/16/2020 | Vox |
| TX | San Antonio | University Hospital | 7/15/2020 | News 4 San Antonio |
| TX | San Antonio | University of Texas Health Science Center | 7/12/2020 | CNN |
| VA | Chesapeake | Chesapeake Regional Care | 7/29/2020 | 13 News Now |
| VA | Newport News | Riverside Health System | 7/29/2020 | 13 News Now |
We also analyzed HHS distribution of remdesivir. Initially, the allocation criteria was based on the proportion of total hospitalized COVID-19 patients, but the agency revised it on July 15th to prioritize proportion of new hospitalized COVID-19 patients over the past seven days.[34] HHS has not made public the data it uses to make these decisions.
On August 13th, we constructed an alternate database, using data compiled from state officials by the COVID Tracking Project.[35] We focused on two critical variables purportedly used by HHS: total COVID-19 hospitalizations and new COVID-19 hospitalizations.[36] Because of data limitations, we excluded some states from the analysis.[37] State data quality varied considerably.[38] We compiled remdesivir distribution data from the HHS website, beginning from the date HHS implemented its new distribution policy (Weeks 2 – 6). HHS reports remdesivir data in cases.[39] The full database is available online.[40]
We find that the hospital burden does not appear to explain the relative allocation of remdesivir. States that had large epidemics with more new hospitalizations often received less remdesivir.
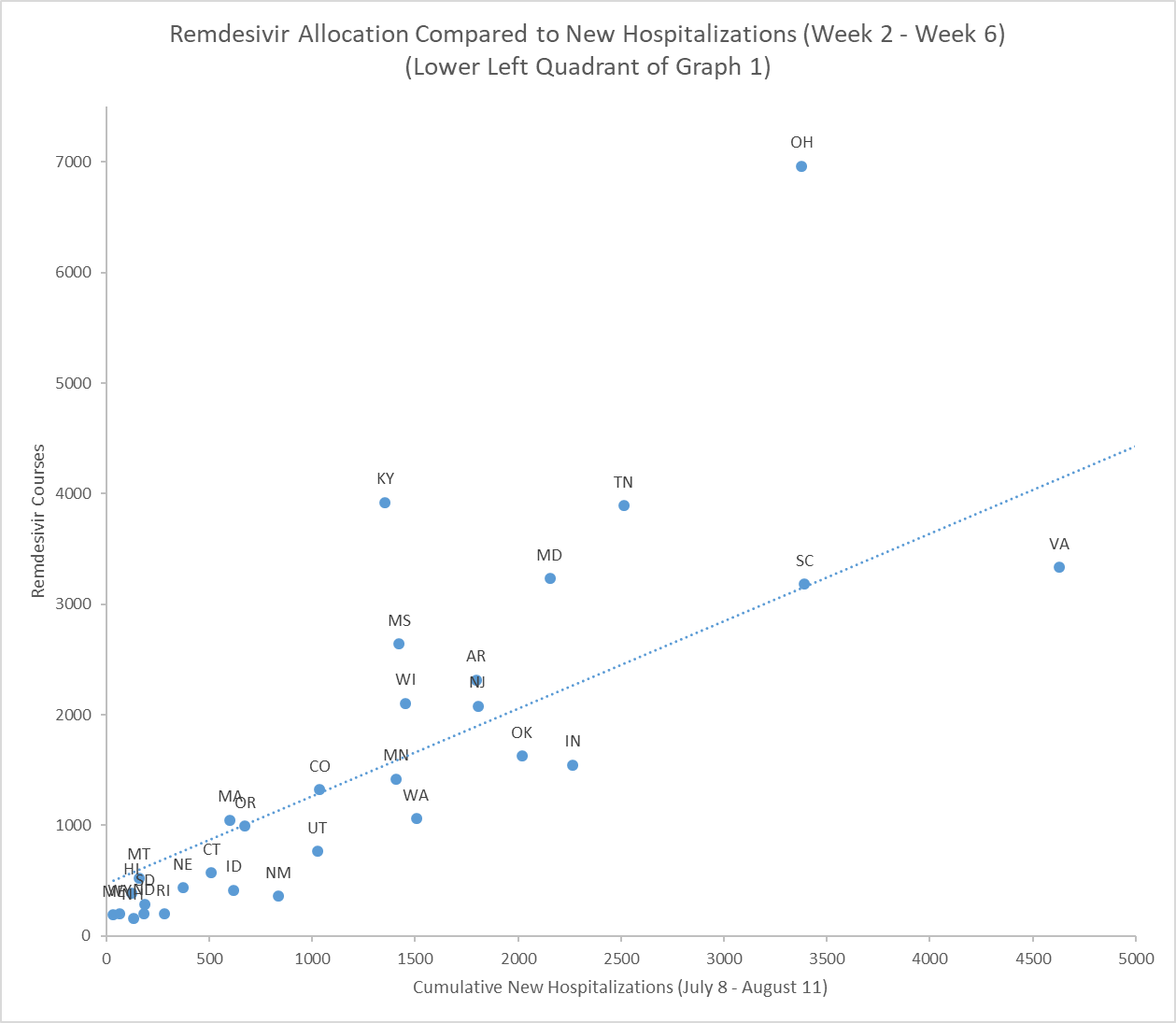
The graphs below start to illustrate the difference between HHS policies and action.[41] Across the five weeks analyzed, Alabama, Arizona, Georgia, South Carolina and Virginia received less remdesivir than Ohio, despite having more new hospitalizations.[42]
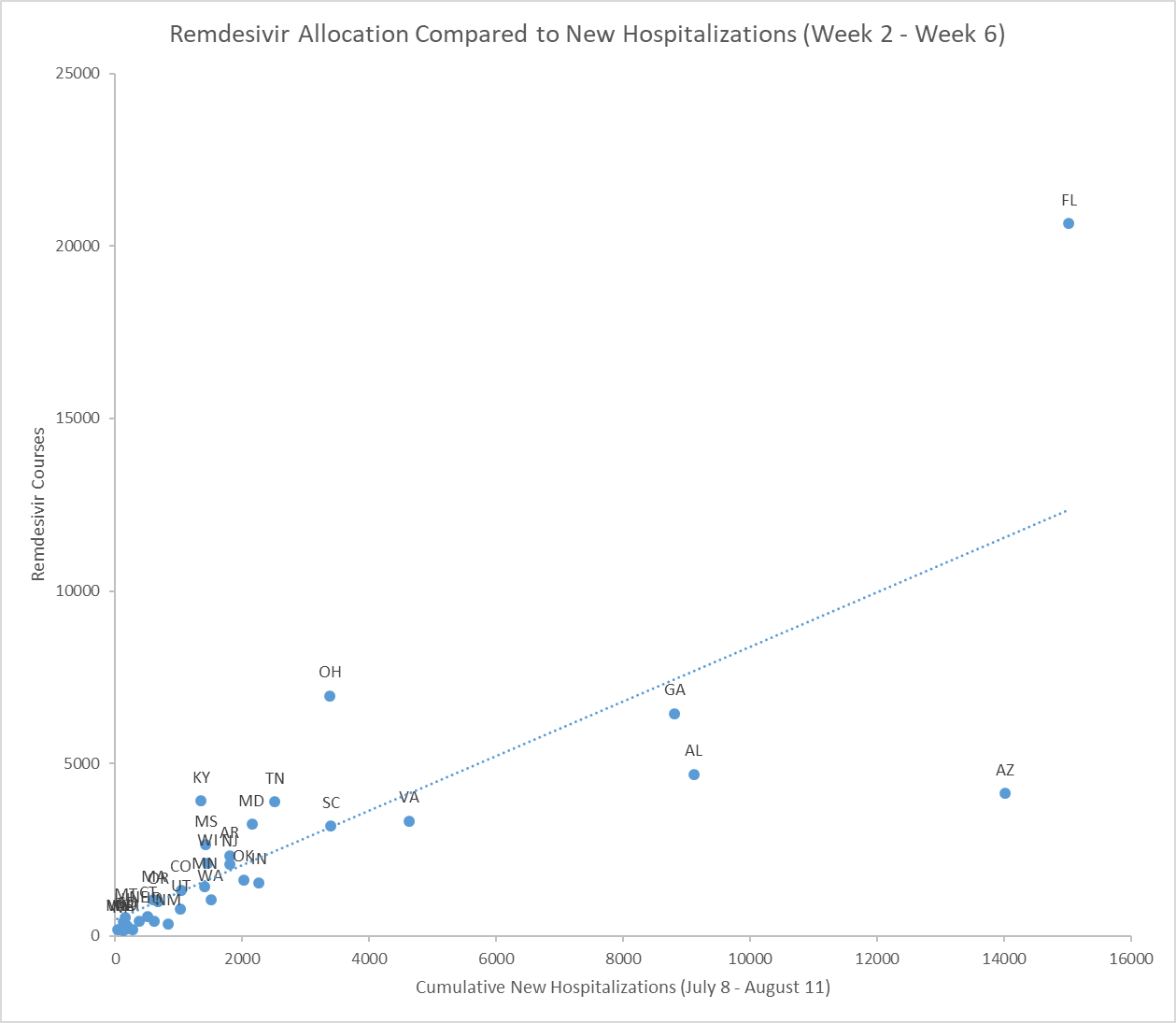
Kentucky also received more remdesivir than eleven other states with more new hospitalizations.
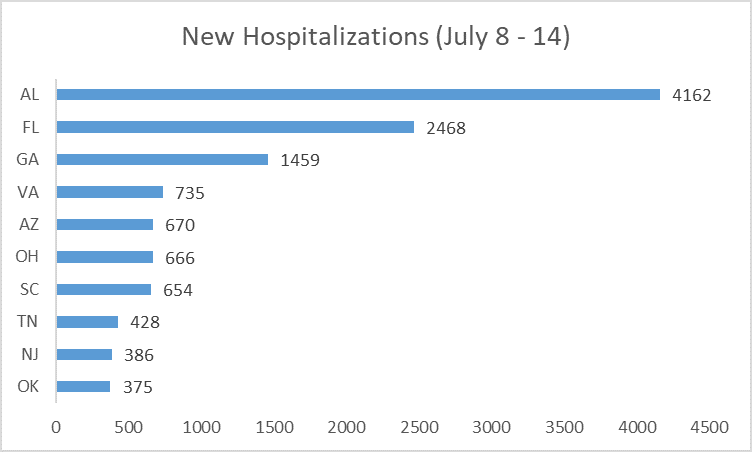
The second distribution week provides the clearest example of HHS’ failure to follow its own policies. The allocation decision was likely made on July 15 based on data from the preceding seven days (July 8 to 14).[43] Of the states that publicly reported new hospitalizations, Alabama, Florida and Georgia reported the most.[44]
Among the states with many new hospitalizations, some also had many people already hospitalized at that moment—including Florida, Georgia and Alabama.
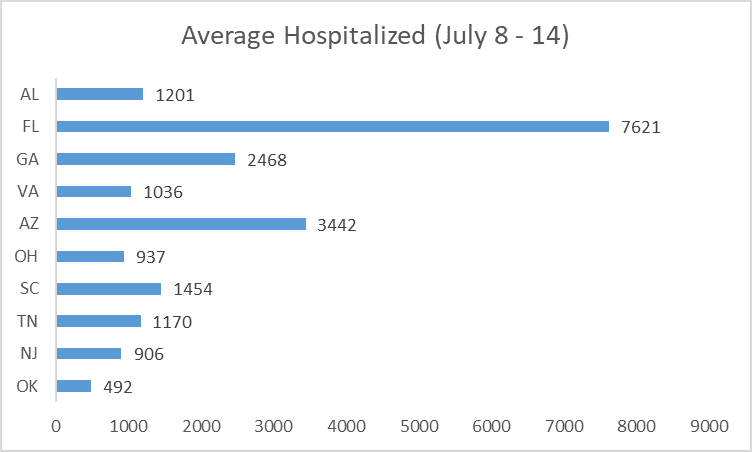
But remdesivir allocation did not reflect these two factors. Georgia was not allocated any courses that week. Ohio received more than Alabama, Arizona, Georgia, and Virginia—even though it had fewer new admissions and fewer total hospitalized patients. Florida received a substantial amount, after Senator Marco Rubio (R-Florida) publicly complained the week before.[45]
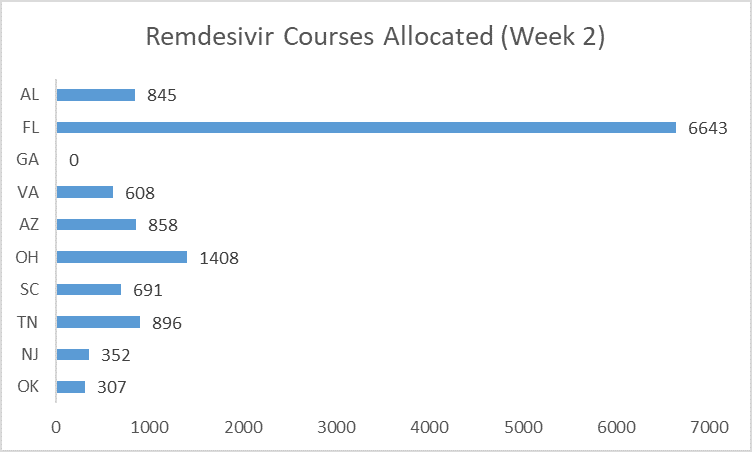
HHS has repeatedly failed to allocate remdesivir based on its own stated criteria. Other egregious failures include:
- Ohio over Georgia: From July 8th until August 4th, Ohio recorded 2736 new hospital admissions. The total people hospitalized on a given day hovered around 1000. Georgia, on the other hand, had 7200 new hospital admissions over the same time period. The total people hospitalized on a given day was around 2500 to 3000. Ohio was allocated 5043 courses of remdesivir.[46] Georgia was allocated 3334;[47]
- Maryland over Alabama: Based on data from July 22nd to August 4th, HHS distributed more than 450 treatment courses of remdesivir to Maryland over Alabama—even though Alabama had nearly three times as many people hospitalized and new admissions;[48] and
- Flooding Montana: From July 8th to August 11th, Montana reported 159 new hospitalizations. HHS, however, allocated the state nearly 525 treatment courses, even as other states were reporting shortages.[49]
Gilead’s lack of supply and the Trump Administration’s incompetence or malfeasance has seriously limited access to remdesivir in the U.S.
Global Shortages
Gilead recently gave permission to some generic manufacturers to sell remdesivir in some low- and middle-income countries.[50] But the licensing agreements are narrow, restricted to a fixed number of countries and companies, and exclude much of the global population.[51] The terms are secret.
For the countries excluded from the agreements, Gilead may be the sole supplier. This is particularly concerning given that Gilead also recently announced deals with the U.S. and the E.U. for nearly all of its supply in June and July. As we described previously, Gilead will likely have only 15,300 courses not committed to the U.S. or E.U. that are available until the end of September.[52] Global access may be seriously constrained.
To estimate the global shortage, we estimated the number of patients who may be eligible for treatment with remdesivir (under the criteria established in the FDA’s original emergency use authorization for the drug[53]) in countries excluded from Gilead’s voluntary license and deals. First, we collected projections by the Institute for Health Metrics and Evaluation for hospital admissions from August to September.[54] Second, we estimated the percentage of hospitalized patients who may eligible for treatment with remdesivir. The treatment is currently authorized for severe COVID-19, which the FDA defines as “patients with low blood oxygen levels or needing oxygen therapy or more intensive breathing support such as a mechanical ventilator.”[55] We used the International Severe Acute Respiratory and Emerging Infections Consortium dataset —which includes more than 80,000 cases across 42 countries—to estimate the proportion of hospitalized cases that “received some degree of oxygen supplementation” as a proxy for severe COVID-19 (66.9% of hospitalized cases).[56]
The results are presented in the table below. We estimate there will be 602,106 patients hospitalized with severe COVID-19 in the excluded countries from August to September. By the end of September, we thus project there will have been hundreds of thousands of hospitalized patients in the excluded countries potentially eligible for treatment with remdesivir.
Table 3: COVID-19 Projections in Countries with Limited Remdesivir Access
| Country | Patients Admitted to Hospital | Severe COVID-19 (Hospitalized Patients Requiring Supplemental Oxygen) | ||
| August | September | August | September | |
| Albania | 566.5 | 570.1 | 379.0 | 381.4 |
| Andorra | 2.3 | 3.5 | 1.5 | 2.3 |
| Argentina | 18,276.7 | 27,701.5 | 12,227.1 | 18,532.3 |
| Australia | 1,778.9 | 5,110.8 | 1,190.1 | 3,419.1 |
| Bahrain | 343.2 | 177.9 | 229.6 | 119.0 |
| Bolivia | 14,962.3 | 12,096.8 | 10,009.8 | 8,092.7 |
| Brazil | 189,442.5 | 101,298.8 | 126,737.0 | 67,768.9 |
| Canada | 624.5 | 479.1 | 417.8 | 320.5 |
| Chile | 9,248.1 | 6,975.9 | 6,187.0 | 4,666.9 |
| Colombia | 47,239.4 | 22,605.7 | 31,603.2 | 15,123.2 |
| Ecuador | 17,012.3 | 16,124.4 | 11,381.2 | 10,787.2 |
| Iran | 38,864.6 | 51,825.6 | 26,000.4 | 34,671.3 |
| Iraq | 18,230.3 | 20,411.5 | 12,196.1 | 13,655.3 |
| Israel | 1,521.7 | 2,848.2 | 1,018.0 | 1,905.4 |
| Japan | 85.0 | 45.8 | 56.8 | 30.7 |
| Jordan | 4.2 | 0.2 | 2.8 | 0.2 |
| Kuwait | 1,342.7 | 2,336.7 | 898.3 | 1,563.3 |
| Lebanon | 246.2 | 142.2 | 164.7 | 95.1 |
| Lithuania | 1.0 | 0.1 | 0.7 | 0.0 |
| Luxembourg | 40.3 | 21.0 | 27.0 | 14.1 |
| Malaysia | 14.0 | 0.9 | 9.3 | 0.6 |
| Malta | 0.2 | 0.0 | 0.1 | 0.0 |
| Mexico | 89,712.6 | 70,641.6 | 60,017.7 | 47,259.2 |
| Montenegro | 92.1 | 285.0 | 61.6 | 190.7 |
| New Zealand | 0.4 | 0.0 | 0.3 | 0.0 |
| North Macedonia | 549.5 | 491.5 | 367.6 | 328.8 |
| Oman | 2,508.2 | 2,647.1 | 1,678.0 | 1,770.9 |
| Paraguay | 250.9 | 291.8 | 167.9 | 195.2 |
| Peru | 43,810.0 | 13,413.9 | 29,308.9 | 8,973.9 |
| Qatar | 356.3 | 346.5 | 238.3 | 231.8 |
| Republic of Korea | 21.7 | 1.3 | 14.5 | 0.9 |
| Russian Federation | 11,613.4 | 12,929.0 | 7,769.4 | 8,649.5 |
| San Marino | 0.8 | 0.8 | 0.5 | 0.5 |
| Saudi Arabia | 5,935.7 | 4,167.6 | 3,971.0 | 2,788.1 |
| Serbia | 537.6 | 371.3 | 359.7 | 248.4 |
| Singapore | 1.4 | 0.1 | 0.9 | 0.1 |
| Syrian Arab Republic | 250.0 | 1,136.5 | 167.2 | 760.3 |
| Turkey | 2,170.3 | 2,469.2 | 1,451.9 | 1,651.9 |
| United Arab Emirates | 326.6 | 307.3 | 218.5 | 205.6 |
| Uruguay | 6.5 | 0.4 | 4.4 | 0.3 |
| Venezuela | 774.7 | 549.2 | 518.3 | 367.4 |
| Yemen | 300.7 | 115.5 | 201.2 | 77.3 |
| Subtotal | 519,066.6 | 380,942.3 | 347,255.5 | 254,850.4 |
| Total | 900,009 | 602,106 | ||
Making Medicines Work
No one corporation can supply the world with a COVID-19 treatment or vaccine. Although remdesivir’s efficacy remains in question, the remdesivir story shows the critical need for multiple suppliers to help scale-up production for COVID-19 treatments and vaccines. Had other suppliers been permitted to produce remdesivir, the U.S. may not have faced serious shortages of the experimental treatment once it was authorized for emergency use.
Governments around the world should permit generic suppliers for COVID-19 treatments and vaccines, so if such treatments and vaccines receive the appropriate regulatory authorization or approval, they can be rapidly deployed. (Vaccines should not be allowed through an emergency use authorization.). In the U.S., Secretary Azar has existing executive authority that could allow him to increase supply and lower prices. Under Section 1498, Secretary Azar can authorize low-cost generic suppliers for patented medicines in exchange for a reasonable royalty.
The remdesivir case illustrates how powerful this authority can be. Suppliers in China, India and Bangladesh have already begun production of remdesivir, and demand from the U.S. could drive further investments in production. In a historic move, a bipartisan group of 34 state and territory attorneys general recently urged Secretary Azar to break Gilead’s monopoly and authorize generic competition for remdesivir. Secretary Azar should heed this call, so Gilead’s monopoly does not constrain supply or facilitate price gouging.
(Emergency use authorization is not the same as regulatory approval. The FDA may eventually not approve remdesivir based on a determination by agency experts that there is not substantial evidence that the drug is effective or has a favorable benefit-risk profile. In any case, clinical need and scientific evidence should guide its use—not supply barriers caused by monopoly control on medicines.)
For equitable distribution of remdesivir, Secretary Azar should increase transparency around allocation mechanisms. All data and models used to support medicine allocation should be publicly available. Allowing external observers to verify the basis for medicine allocation is critical for government accountability.
Around the world, governments should also exercise their patent use authorities to authorize generic competition and, if applicable, increase allocation transparency.[57] As we noted in People over Patents, in response to COVID-19, some governments have streamlined processes for the government use of patented inventions.[58] Canada, Germany and Indonesia have adopted new non-voluntary patent licensing policies. Others have taken steps to license patents, with Chile and Ecuador calling for licenses and Israel issuing a license for another candidate COVID-19 treatment.
Remdesivir shortages are only a harbinger. Meeting global demand for medicines authorized by regulatory authorities during a pandemic will require breaking monopoly control over medicines.
[1] Gilead’s Investigational Antiviral Remdesivir Receives U.S. Food and Drug Administration Emergency Use Authorization for the Treatment of COVID-19, Gilead (May 1, 2020), https://tinyurl.com/y9w53ya4. Australia, Canada, the European Union, India, Israel, Japan, Singapore, South Korea and Taiwan have also authorized emergency use of remdesivir.
[2] Who Gets the Covid-19 Vaccine First? Here’s One Idea, NY Times (July 23, 2020), https://tinyurl.com/y4x4zdy8
[3] Sen. Marco Rubio, Twitter (July 16, 2020), https://tinyurl.com/y5h77yzp The state received twice as much the week after.
[4] Bay Area Hospitals Say Remdesivir is in Short Supply, KQED (July 22, 2020), https://tinyurl.com/yyyr9ua2
[5] Because we rely exclusively on publicly available sources (i.e., news articles), this is likely an underestimate.
[6] Manufacturers have begun production in Bangladesh, China and India. Exclusive: Bangladesh’s Beximco to begin producing COVID-19 drug remdesivir – COO, Reuters (May 5, 2020), https://tinyurl.com/yckfypwn. A Chinese company is already mass producing Gilead’s experimental coronavirus drug, Endpoint News (February 12, 2020), https://tinyurl.com/rq236pl. India starts producing generic Remdesivir amid pandemic, Anadolu Agency (June 22, 2020), https://tinyurl.com/yyenrr8c. The U.S. signaling that it would consider purchasing from generic producers could facilitate a rapid scale-up in producers.
[7] See Domestic Shortages.
[8] Voluntary Licensing Agreements for Remdesivir, Gilead, https://tinyurl.com/y5bey5ue
[9] See Tracking Supply.
[10] See Global Shortages.
[11] Gilead Sciences Form 10-Q, (June 30, 2020), http://investors.gilead.com/static-files/5648e7dd-c981-4d5b-8c8f-22342d60a946 (“Further, given that COVID-19 has been designated as a pandemic and represents an urgent public health crisis, and given that there is no assurance that we will be able to meet global supply needs for remdesivir, we have observed and are likely to continue to face significant public attention and scrutiny over the complex decisions made regarding the allocation, business models and pricing decisions with respect to remdesivir.”).
[12] The Real Story of Remdesivir, Public Citizen (May 7, 2020), https://www.citizen.org/article/the-real-story-of-remdesivir/
[13] The U.S. Government’s Apparent Co-Ownership of Patents Protecting Remdesivir, PrEP4ALL-NYU School of Law (May 20, 2020), https://tinyurl.com/yy9nkrma
[14] An Open Letter from Daniel O’Day, Chairman & CEO, Gilead Sciences, Gilead (June 29, 2020), https://tinyurl.com/y9vd4znu
[15] Andrew Hill et al,, Minimum Costs to Manufacture New Treatments for COVID-19, Journal of Virus Eradication (April 6, 2020). https://tinyurl.com/yyjnybeb.
[16] Ameet Sarpatwari et al., Missed Opportunities on Emergency Remdesivir Use, JAMA (June 24, 2020), https://jamanetwork.com/journals/jama/fullarticle/2767752.
[17] Beigel et al., Remdesivir for the Treatment of Covid-19 — Preliminary Report, The New England Journal of Medicine (May 22, 2020), https://tinyurl.com/y7up8k42
[18] Trump Administration Secures New Supplies of Remdesivir for the United States, HHS (June 29, 2020), https://tinyurl.com/yc6km7bz
[19] European Commission secures EU access to Remdesivir for treatment of COVID-19, European Commission (July 29, 2020), https://tinyurl.com/y48rj4b7
[20] 547,200–30,000–501,900=15,300
[21] H1 & Q2 2020 Earnings Results, Gilead (July 30, 2020), https://tinyurl.com/y5o3xxmq. Pg. 7.
[22] Gilead Sciences (GILD) Q2 2020 Earnings Call Transcript, Gilead Sciences Earnings Call (June 30, 2020), https://tinyurl.com/y3bskbcr (“We should be in a position to have cumulatively of over 2 million treatment courses. And some of those, obviously, were used during the donation and some are being used in Q3.”) Gilead says it has donated 1.5 million vials. At 6.25 vials per course, that represents 240,000 courses. H1 & Q2 2020 Earnings Results, Gilead (July 30, 2020), https://tinyurl.com/y5o3xxmq. Courses can range from 5 to 10 days depending on disease severity under the EUA.
[23] Pfizer Announces Agreement With Gilead To Manufacture Remdesivir For Treatment Of Covid-19, Pfizer (August 7, 2020), https://tinyurl.com/yykg4dyp. UK’s Hikma making Gilead’s COVID-19 drug remdesivir to increase supply, Reuters (August 7, 2020), https://tinyurl.com/y3k2c86t
[24] Gilead Sciences Form 10-Q, (June 30, 2020), http://investors.gilead.com/static-files/5648e7dd-c981-4d5b-8c8f-22342d60a946 (“Further, given that COVID-19 has been designated as a pandemic and represents an urgent public health crisis, and given that there is no assurance that we will be able to meet global supply needs for remdesivir, we have observed and are likely to continue to face significant public attention and scrutiny over the complex decisions made regarding the allocation, business models and pricing decisions with respect to remdesivir.”).
[25] Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment, U.S. FDA (May 1, 2020), https://tinyurl.com/y85wje8a
[26] COVID-19 Treatment Guidelines: Remdesivir, NIH (July 24, 2020), https://tinyurl.com/y5bphjel
[27] CFR – Code of Federal Regulations Title 21, U.S. FDA (April 1, 2019), https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=314.81
[28] A drug everyone wants: how local hospitals are using Remdesivir, (July 29, 2020), 13 News Now
[29] Pinellas County Commission pleads with DeSantis to send coronavirus drug, (July 27, 2020), Tampa Bay Times
[30] I-Team: Small shipments of Remdesivir continue as Nevada faces short supply (Aug 5, 2020), Channel 8 News Now Las Vegas. Each case contains 40 vials. A treatment course, on average, is 6.25 vials. Allocation of Commercially Available Remdesivir by Jurisdiction, HHS, https://tinyurl.com/y4zknrpr
[31] S.C. hospitals cope with shortage of promising COVID-19 drug, (July 31, 2020), WCSC
[32] One article reported that the entire hospital system (Baptist Health) had run out.
[33] If a city is reporting a shortage, then we inferred that at least one hospital in the city is reporting a shortage as well.
[34] We note that the HHS website is still somewhat ambiguous but it now provides “The current methodology uses the same proportional strategy as before; however, slightly different data elements are used.. Instead of basing the allocations on the proportion of all admitted COVID-19 patients (confirmed or suspected), whether old or new patients, the allocations are now based on a state’s/territory’s proportion of newly admitted COVID-19 patents (confirmed or suspected over the previous seven-day period). Remdesivir for the Commercial Market FAQ, HHS, https://tinyurl.com/y3lydrrk
[35] The COVID Tracking Project makes its data available under a Creative Commons CC BY-NC-4.0 license. https://covidtracking.com/.
[36] We assessed total hospitalizations and new hospitalizations as the most important variables. All other things being equal, we did not expect significant variation in ICU admissions. Remdesivir for the Commercial Market FAQ, HHS, https://tinyurl.com/y3lydrrk (“The data fields specifically used for determining the allocation of remdesivir include: Previous day’s new adult admissions for confirmed COVID-19; Previous day’s new adult admissions for suspected COVID-19; Total adults hospitalized for COVID-19 (suspected and confirmed); Total hospitalized for COVID-19 (confirmed only); Total adults in Intensive Care Unit with COVID-19 (suspected and confirmed); Total adults in Intensive Care Unit with COVID-19 (confirmed).”)
[37] AK, CA, DE, IA, IL, LA, MI, MO, NC, NV, NY, PA, TX, VT and WV did not report more detailed hospitalization data and were excluded. KS did not report the currently hospitalized for one week and was excluded.
[38] Some states, for example, changed their reporting methodology, leading to data spikes. Others had large increases likely attributable to reporting delays. The analysis could be stronger had HHS made its data publicly available.
[39] Each case contains 40 vials. A treatment course, on average, is 6.25 vials. Allocation of Commercially Available Remdesivir by Jurisdiction, HHS, https://tinyurl.com/y4zknrpr
[40] Public Citizen, Remdesivir Distribution Database, https://www.citizen.org/wp-content/uploads/Remdesivir-distribution-database.xlsx
[41] This is an imperfect measure because we combined data on remdesivir allocation across multiple weeks—as opposed to comparing week by week. We also focused exclusively on new hospitalizations.
[42] State data quality varied considerably. For example, there was an unexplained spike in Arizona on August 7th. The analysis could be stronger had HHS made its data publicly available.
[43] Remdesivir for the Commercial Market FAQ, HHS, https://tinyurl.com/y3lydrrk (“As of July 15, 2020, HHS will utilize information that hospitals input into the TeleTracking system and the HHS Protect platform on a daily basis to determine allocations for each distribution period for commercially available remdesivir. Allocations are determined on Wednesdays of each distribution week and are based on hospital data input into TeleTracking/HHS Protect over the previous seven days.”)
[44] Some states were excluded from our analysis because they do not publicly report complete data about hospitalizations.
[45] Sen. Marco Rubio, Twitter (July 16, 2020), https://tinyurl.com/y5h77yzp. Twice as much remdesivir was sent after the complaint. It went back to close to the original allocation the week after.
[46] 788 cases from week 2 to week 5. Each case has 40 vials. An average treatment course is 6.25 vials.
[47] 521 cases from week 2 to 5. Each case has 40 vials. An average treatment course is 6.25 vials. Georgia finally received a substantial increase in week 6.
[48] See Remdesivir Distribution Database.
[49] 82 Cases. See Remdesivir Distribution Database.
[50] Voluntary Licensing Agreements for Remdesivir, Gilead, https://tinyurl.com/y5bey5ue
[51] Remdesivir Should Be in the Public Domain; Gilead’s Licensing Deal Picks Winners and Losers, Public Citizen (May 12, 2020), https://tinyurl.com/y5cae44v
[52] See Tracking Supply.
[53] Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment, U.S. FDA (May 1, 2020), https://tinyurl.com/y85wje8a
[54] The Institute for Health Metrics and Evaluation makes available data through a Creative Commons Attribution-NonCommercial 4.0 International License. Notably, there was no data available for China. The model also did not have data for Brunei Darussalam, Liechtenstein and Monaco. Those countries were excluded from this analysis. The Institute for Health Metrics and Evaluation, http://www.healthdata.org/
[55] Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment, U.S. FDA (May 1, 2020), https://tinyurl.com/y85wje8a
[56] This is an imperfect proxy. There may also be some selection bias in reported cases. International Severe Acute Respiratory and Emerging Infections Consortium dataset, (July 13, 2020), pg. 2, https://isaric.tghn.org/.
[57] Some governments may not distribute remdesivir themselves.
[58] Public Citizen, People Over Patents (August 10, 2020), https://www.citizen.org/article/people-over-patents/.