mRNA-1273 Vaccine Patent Landscape (For NIH-Moderna Vaccine)
By Mario Gaviria and Burcu Kilic
Safe and effective vaccines are key to combating the Covid-19 pandemic; however, patents and other intellectual property claims directed at vaccine technologies create legal barriers for equitable access and fair allocation. No corporation produces at scale to supply the world. Providing timely global access will depend in significant part on increasing supply, including by transferring technology to qualified manufacturers. Much of this technology is claimed as patented, proprietary, or confidential in nature.
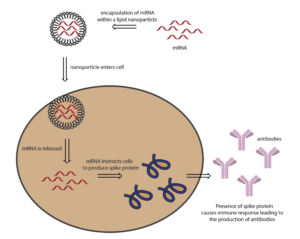
The vaccine candidate co-developed by the U.S. National Institutes of Health (NIH) and Moderna, mRNA-1273 SARS-CoV-2, employs the use of lipid nanoparticle (NP) technology to deliver mRNA to cells. Once the lipid nanoparticle is injected into a patient, it travels into the cells and instructs them to produce the SARS-CoV-2 spike protein. The presence of this coronavirus protein is thought to trigger an immune response leading to the production of antibodies.[1] If the patient is infected with coronavirus, the antibodies will identify and bind to the virus, which triggers a series of events resulting in the elimination of the virus.
mRNA-1273 is in Phase 3 clinical trials. The NIH and Moderna announced promising but preliminary trial results on November 16th.[2]
We identified several patents claimed by Moderna relating to the pertinent vaccine technologies.[3] We placed them in four groups based on their description and their primary independent claim:
- Patents directed at an mRNA vaccine or RNA generally

- Patents directed at Lipids/NP + mRNA
- Patents specifically directed at pharmaceutical compositions involving lipid NP + mRNA.
Below is our non-exhaustive list. In a recent financial statement, Moderna suggested that it relies to a certain extent on trade secrets, know-how, and technology, which are not protected by patents, to maintain its competitive position.[4] Moderna has announced that it will not enforce their patent rights against those making vaccines intended to combat the pandemic.
| Patent/Published Application | Applicant/Assignee | Filing Date | Status | Invention Type |
| US 10,703,789 | Moderna | June 12, 2019 | Active | PC: Lipids/NP + mRNA |
| US 10,702,600 | Moderna | February 28, 2020 | Active | Betacoronavirus mRNA Vaccine |
| US 10,577,403 | Moderna | June 12, 2019 | Active | PC: Lipids/NP + mRNA |
| US 10,442,756 | Moderna | December 18, 2017 | Active | Lipids/NP + mRNA |
| US 10,266,485 | Moderna | June 11, 2018 | Active | Lipids/NP + mRNA |
| US 10,064,959 | Moderna | April 21, 2017 | Active | mRNA synthesis |
| US 9,868,692 | Moderna | March 31, 2017 | Active | Lipids/NP + mRNA |
| US2020/0206362 | Moderna | October 11, 2019 | Pending | PC: Lipids/NP + mRNA |
| US2020/0164038 | Moderna | July 29, 2019 | Pending | PC: Lipids/NP + mRNA |
| US2019/0015501 | Moderna | September 27, 2018 | Pending | Nucleic acid vaccine |
| WO2016/118724 | Moderna | January 21, 2016 | Published | Lipids/NP + mRNA |
| WO2016/118725 | Moderna | January 21, 2016 | Published | Lipids/NP + mRNA |
[1] https://www.modernatx.com/mrna-technology/mrna-platform-enabling-drug-discovery-development
[2] https://investors.modernatx.com/news-releases/news-release-details/modernas-covid-19-vaccine-candidate-meets-its-primary-efficacy
[3] Pharmaceutical companies are not the only claimants of key technology. The U.S. government claims a patent on a key technology which may be relevant for Moderna to stabilize the spike protein. See Public Citizen, Leading COVID-19 Vaccine Candidates Depend on NIH Technology (Nov. 10, 2020), https://www.citizen.org/article/leading-covid-19-vaccines-depend-on-nih-technology/.
[4] “If any trade secret, know-how, or other technology not protected by a patent were to be disclosed to or independently developed by a competitor, our business and financial condition could be materially adversely affected. Failure to obtain and maintain all available regulatory exclusivities and broad patent scope and to maximize patent term restoration or extension on patents covering our products may lead to loss of exclusivity and early biosimilar entry resulting in a loss of market share and/or revenue.” Moderna, Quarterly Report, June 30, 2020 https://www.sec.gov/Archives/edgar/data/1682852/000168285220000017/mrna-20200630.htm