
Paxlovid Patent Landscape
Pfizer's path to building patent barriers in a global pandemic
By Benjamin Wild
Background
Pfizer’s investigational COVID-19 treatment, Paxlovid, is a combination of a new compound, nirmatrelvir (“PF-07321332”), and an old drug, ritonavir.
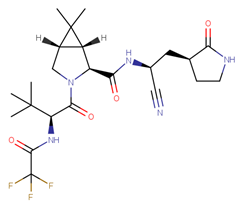 |
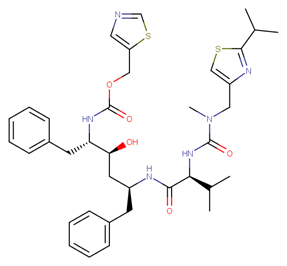 |
| Nirmatrelvir |
Ritonavir |
On Dec. 22, 2021 the treatment received an emergency use authorization in the U.S. for twice-daily administration of nirmatrelvir, dosed at 300mg (two 150mg tablets), and ritonavir, dosed at 100mg (one tablet), for five days in patients at high risk of progressing to severe disease. Similar authorizations and guidance on use have been issued in other jurisdictions such as Bangladesh, the UK, and Europe.
Pfizer says that, compared to placebo, the treatment may reduce the risk of hospitalization or death in high-risk patients by 89% when taken within three days of symptom onset, and by 88% when taken within five days of symptom onset. Full clinical trial results have not yet been made available to the public.
Ritonavir was developed in the late 1980s as an antiretroviral medication for HIV/AIDS treatment. Its main patents (compound, polymorph, etc.) have expired and there are now generic manufacturers of the drug.
Nirmatrelvir, on the other hand, is a new chemical entity that was purportedly developed from the ground up during the COVID-19 pandemic, with the drug discovery process having started days after the World Health Organization declared that COVID-19 could be characterized as a pandemic in Mar. 2020. That being said, the foundations for nirmatrelvir’s development were laid in 2003 when Pfizer was working on an antiviral drug against SARS-CoV-1 (a protease inhibitor dubbed “PF-00835231”), but whose development was halted when the SARS outbreak was contained.
The target protease binding site in SARS-CoV-1 is identical to that of SARS-CoV-2, and so Pfizer’s work on “PF-00835231” gave its researchers a good place to start when it came to developing a molecule that would be active against SARS-CoV-2. “PF-00835231” does not have oral bioavailability, which makes administering the drug more burdensome than furnishing patients with a treatment in pill form. To arrive at a molecule which could be taken orally to act against SARS-CoV-2, several significant structural modifications were made to arrive at nirmatrelvir.
Given how recently nirmatrelvir was developed, the patent landscape relating to the molecule and the Paxlovid treatment is relatively sparse at present. When patent landscapes are not mature, generic competitors face uncertainty regarding what patent protection may be obtained, and in which jurisdictions, for a given pharmaceutical product, hindering their ability to plan, with potential consequences for supplying global markets.
Patent Filing Process Overview
Filing an “international” Patent Cooperation Treaty (PCT) application is common at the 12-month point as the PCT system gives the applicant access to 153 contracting states, which represent most (but, importantly, not all) of the world’s countries, and pushes back the date at which it is required to file national/regional applications until after the applicant has had sight of an examiner’s opinion of the claimed invention’s patentability (~15/16 months)[ii]. At the 18-month date, the PCT application is published and its contents are made available to the public for the first time[iii]. At or before the 30/31-month dates, applicants file their desired national and regional patent applications[iv]. The claimed invention’s patentability will be assessed by each relevant national/regional patent office according to their assessment criteria[v]; during the national/regional phase, it is usual for amendments to be made to the claims and for it to take between 2-5 years to reach grant or refusal. Even if a patent application is granted, the granted patent may be challenged resulting in amendment, revocation, or no change to its scope. Granted patents are negative rights that allow the owner to restrict others from making, using, or selling a given invention; being granted a patent does not automatically give the owner the right to make, use, or sell their claimed invention as it is possible that there are other patent rights which prevent them from doing so. The PCT filing date is the date from which the usual 20-year patent term is calculated for national/regional patent applications derived from the PCT application[vi]; however, this term can be more in some jurisdictions depending on whether patent term extensions are available. |
Patent Landscape
At present, much of what we know about Paxlovid’s patent landscape comes from a voluntary license agreement between Medicines Patent Pool (MPP) and Pfizer. This was devised to facilitate the granting of sub-licenses to qualified generic manufacturers in 95 countries (mostly low- and lower-middle-income countries) for the production of generic versions of Paxlovid.
Exhibit B of the MPP-Pfizer license agreement details that Pfizer has only one patent family related to the Paxlovid product which, as of this writing, has no granted patents. The “international” PCT application PCT/IB2021/057281 listed was expected to publish in Mar. 2022 (18 months from the earliest priority date of Sep. 3, 2020 – see the four “inactive” U.S. patent applications mentioned in Exhibit B). However, Pfizer requested early publication of the PCT application; the application’s contents were made available to the public on Dec. 16, 2021 with a publication number of WO/2021/250648.
Pfizer has indicated, in Exhibit B, that it has filed, or intends to file, patent applications in 61 countries and four regional patent offices. The above map illustrates the potential patent coverage from these filings (interactive version of the map may be found here).
Of the applications listed in Exhibit B, it appears that there are already pending patent applications in 34 countries and with two regional patent offices[vii] based on the application numbers mentioned. Inspecting the list of national phase entries mentioned on the online register for WO/2021/250648 reveals that there are further pending patent applications in an additional five countries and one regional patent office[viii]. The remaining applications listed are to be filed via the PCT system (PCT/IB2021/057281) or using registration systems for jurisdictions such as Hong Kong and Macau[ix]. There also are a handful of jurisdictions which Pfizer did not mention in the license agreement which are still available to them via their pending PCT application[x].
| Indication that applications have been filed | Indication in Exhibit B that applications will be filed | Additionally accessible via pending PCT application | |
| National | Argentina, Aruba, Australia, Bangladesh, Bolivia, Brazil, Canada, Chile, Colombia, Costa Rica, Cuba, Dominican Republic, Georgia, Guatemala, Honduras, India, Israel, Kosovo, Lebanon, Mexico, Mongolia, New Zealand, Nicaragua, Nigeria, Pakistan, Panama, Paraguay, Peru, Philippines, Republic of Korea, Russian Federation, Saudi Arabia, Singapore, Taiwan, Ukraine, United Arab Emirates, United States of America, Uruguay, Venezuela (Bolivarian Republic of) | Algeria, Bahamas, Bahrain, Barbados, China, Ecuador, Egypt, El Salvador, Hong Kong [registration via Chinese, European, or UK patent application], Indonesia, Japan, Kuwait, Macao [registration via Chinese patent application], Malaysia, Oman, Qatar, South Africa, Sri Lanka, Thailand, Trinidad and Tobago, Uzbekistan, Vietnam | Angola, Antigua and Barbuda, Belize, Brunei Darussalam, Democratic People’s Republic of Korea, Djibouti, Dominica, Grenada, Iran (Islamic Republic of), Jordan, Lao People’s Democratic Republic, Libya, Madagascar, Papua New Guinea, Saint Kitts and Nevis, Saint Lucia, Saint Vincent and the Grenadines, Samoa, Seychelles, Syrian Arab Republic |
| Regional | ARIPO (Botswana, Eswatini, Gambia, Ghana, Kenya, Lesotho, Liberia, Malawi, Mozambique, Namibia, Rwanda, São Tomé and Principe, Sierra Leone, Sudan, Tanzania, Uganda, Zambia, Zimbabwe), EAPO (Armenia, Azerbaijan, Belarus, Kazakhstan, Kyrgyz Republic, Tajikistan, Turkmenistan, Russian Federation), EPO (Albania, Austria, Belgium, Bosnia and Herzegovina [extension state requested], Bulgaria, Cambodia [validation state not yet requested], Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Italy, Latvia, Liechtenstein, Lithuania, Luxembourg, Malta, Moldova [validation state requested], Monaco, Montenegro [extension state requested], Morocco [validation state requested], Netherlands, North Macedonia, Norway, Poland, Portugal, Romania, San Marino, Serbia, Slovak Republic, Slovenia, Spain, Sweden, Switzerland, Tunisia [validation state requested], Turkey, United Kingdom) | OAPI (Benin, Burkina Faso, Cameroon, Central African Republic, Chad, Comoros, Congo, Côte d’Ivoire, Equatorial Guinea, Gabon, Guinea, Guinea-Bissau, Mali, Mauritania, Niger, Senegal, Togo) | None |
Given that Sep. 3, 2020 is the earliest priority date for this family, the international phase will end Mar./Apr. 3, 2023 (i.e., 30/31 months from the earliest priority date) for those jurisdictions in which the national or regional phase has not already been entered.
The full list of jurisdictions in which Pfizer intends to pursue patent protection with respect to the family listed in Exhibit B will remain unclear at least until the international phase ends (it will likely take some time for national or regional patent registers to publish details of an associated application having been filed), as does whether grants will be achieved in those jurisdictions.
As mentioned in the background section, it typically takes somewhere between 2-5 years to reach grant or refusal once the national/regional phase has been entered and it is usual for amendments to be made during prosecution; however, it is likely that Pfizer will be using expedited processing in jurisdictions where such a mechanism is available to obtain a faster decision. If there are any patents granted, it is possible for third parties to file challenges, which could result in further amendment, revocation, or no change to the scope of the patent.
For patents that may be granted in this family, the expected expiry date of such granted patents would be Aug. 5, 2041 based on the PCT application filing date of Aug. 6, 2021. This does not include patent term extensions, which may be possible in some jurisdictions.
Pfizer’s filing of applications in 11 non-PCT-accessible countries[xi], it entering the national/regional phase early and/or filing national/regional applications directly in 31 jurisdictions[xii] which are accessible via the PCT system, and it requesting early publication of the PCT application suggests that it will aggressively pursue patent protection for its new compound and treatment.
PCT Application Overview (WO/2021/250648)
 As mentioned above, Pfizer requested early publication of the “international” PCT application listed in Exhibit B (i.e., PCT/IB2021/057281), meaning it is now possible to view the application’s contents and the associated file wrapper.
As mentioned above, Pfizer requested early publication of the “international” PCT application listed in Exhibit B (i.e., PCT/IB2021/057281), meaning it is now possible to view the application’s contents and the associated file wrapper.
There are several reasons why an applicant may elect to have their application publish early, including i) to create prior art issues for other parties that may attempt to obtain patent protection for related subject-matter, ii) to put competitors on notice as to what scope of protection they are intending to pursue, and/or iii) to increase the level of pre-grant damages awarded from potential infringers (it is possible to obtain damages from infringers for the period between when an application is published and granted).
From WO/2021/250648’s bibliographic data, we are informed that the “international” PCT application was filed by Pfizer Inc. [US], that it claims the priority of four earlier US applications (the US applications listed in Exhibit B as “inactive”), with an earliest priority date of Sep. 3, 2020, and that it names nine inventors.
A review of the filing forms for each of the four priority applications and the PCT application confirmed that the same applicant was named in each instance (i.e., Pfizer Inc. [US]) and, while the named inventors varied between applications, it appears that assignments from the inventors to Pfizer were secured prior to the PCT application’s filing date to avoid ownership issues (although we have not yet had sight of the assignment documents themselves).
The filing dates of the relevant priority applications appear to align with key dates in the nirmatrelvir development timeline, as is typical. For example, the first priority application was filed two days after the inventors found that nirmatrelvir not only had good SARS-CoV-2 antiviral activity but also oral activity, and the third priority application was filed a few days before Pfizer announced the drug candidate and its structure at the American Chemical Society Spring 2021 meeting.
The main claim (claim 1) of the PCT application is based on the below Markush, Formula I’’, first described in the third priority document (“P3”).
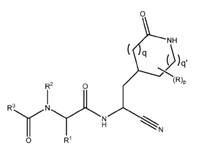 |
| Formula I’’ |
Subsequent claims narrow in scope around nirmatrelvir. Further claims focus on methods of treating coronavirus infections, preparations of medicaments, combination treatments (e.g., with ritonavir), dosage regimens, and so forth; these are the typical array of claim types that are expected to facilitate prosecution of a pharmaceutical patent in various jurisdictions around the world.
The International Searching Authority (ISA) has assessed this PCT application’s patentability and it is their opinion that the subject-matter of all the claims are novel (new); while claim 1, directed to compounds of Formula I’’, has been deemed too broad for an inventive step to be acknowledged, the list of more narrowly defined Markush structures in claim 2 have been deemed to be inventive (along with the claims that are more narrowly defined and are dependent on claim 2).
The written opinion of the ISA is not binding on its contracting states. However, it does provide a breakdown of relevant prior art documents and an assessment of whether patentable subject-matter has been claimed; this is useful for both the applicant and third parties when determining the likelihood of granted patents being obtained.
To achieve speedy grants nationally or regionally, it is likely that Pfizer will limit the claims to the scope of claim 2, given the positive opinion by the ISA, and may pursue broader protection in divisional applications. In fact, from inspection of the European patent register, it can be seen that Pfizer have already pre-emptively limited the claims for the associated European patent application in this manner in preparation for prosecution.
In summary, it would appear likely that granted claims directed to nirmatrelvir and Paxlovid are achievable. Please see Annex 1 for a more detailed on this PCT application.
Other Patent Applications
Results from a search on lens.org for “PF-07321332”[xiii], which was nirmatrelvir’s working name until around Dec. 2021, showed seven published international applications mentioning “PF-07321332” in lists of anti-viral agents/protease inhibitors, five of which also mention ritonavir (outlined in red below). These applications are in the international phase and have not been granted nationally/regionally.
| Title | Identifiers | Priority | Filed | Published | Applicants |
| Compound and method for the treatment of coronaviruses | WO 2021/250038 A1 | Jun. 10, 2020 | Jun. 8, 2021 | Dec. 16, 2021 | Apteeus |
| System, method and use of a certain medication for reducing viral replication in the airways mucosae | WO 2021/234668 A1 | May. 22, 2020 | May. 21, 2021 | Nov. 25, 2021 | Riveros Carlos Alberto |
| Compositions and methods for increasing efficacy of a drug | WO 2021/231872 A1 | May. 15, 2020 | May. 14, 2021 | Nov. 18, 2021 | Healion Bio Inc |
| Products of manufacture and methods for treating, ameliorating or preventing microbial infections | WO 2021/212183 A1 | Apr. 24, 2020 | Apr. 23, 2021 | Oct. 28, 2021 | Centre for Digestive Diseases Pty Ltd |
| Vidofludimus for use in the treatment or prevention of viral diseases | WO 2021/214033 A1 | Apr. 21, 2020 | Apr. 20, 2021 | Oct. 28, 2021 | Immunic AG |
| Treatment of viral pathologies with exogenous ketones | WO 2021/211609 A1 | Apr. 16, 2020 | Apr. 13, 2021 | Oct. 21, 2021 | Buck Inst Res Aging |
| Small molecule therapeutics for the treatment of viral infections | WO 2021/207632 A1 | Apr. 10, 2020 | Apr. 9, 2021 | Oct. 14, 2021 | Univ California |
For the outlined applications, “PF-07321332” is not mentioned in any of the associated priority documents filed before Pfizer unveiled the compound to the public in early April 2021, and it would appear that, in most of the above cases, mention of the drug was not added until each international application was filed. “PF-07321332” is not the primary focus of these applications, with the drug being mentioned speculatively as a possible combination drug, and these applications are therefore unlikely to create considerable freedom to operate issues for Paxlovid.
The Paxlovid patent landscape is not fully mature given how recently nirmatrelvir was developed. It is likely that follow-on applications will be or have been filed by Pfizer to extend the potential period of protection (combinations, dosage regimens, further uses, polymorphs, etc.). There may be additional patent applications (published or unpublished) not uncovered in our searches with bearing on the Paxlovid patent landscape[xiv].
Compulsory Licenses
For countries not covered by or relying on the MPP-Pfizer license agreement for access to Paxlovid, compulsory licenses, government use, and/or Crown use of intellectual property rights (hereafter “CLs”) can play an important role. Where utilised, CLs are typically issued on granted patents; however, some countries allow CLs to be issued for pending patent applications[xv] and others are exploring the possibility of updating their regulations to this end (e.g., Brazil).
| Jurisdictions, sorted by World Bank income classification, which appear to have regulations which expressly1, or could be read as to tacitly2, allow for compulsory licenses/government use/Crown use of pending patent applications include (and may not be limited to): |
| Low – Madagascar1*
Lower-middle – Algeria1*, Honduras1*, India1*, Nicaragua1*, Tunisia1*, Vietnam2*, Zambia1* Upper-middle – Albania1, Costa Rica1, Dominican Republic1, Guatemala1*, Saint Lucia1, Turkey1 High – Australia1, Chile2, Cyprus1, Czech Republic2, France1, Greece2, Ireland1, Israel1, Latvia2, Malta1, Spain1, United Kingdom1 *Mentioned in Exhibit C of the MPP-Pfizer license agreement (i.e. the “country list”) |
Requests starting the process for obtaining CLs in Chile and the Dominican Republic, where national patent applications appear to have been filed based on the application numbers listed in Exhibit B, have already been filed by the NGOs Innovarte (together with The Chilean Association of Pharmaceutical Chemists and Biochemists) and Knowledge Ecology International, respectively, to facilitate generic market entry.
For jurisdictions which allow CLs to be issued for pending patent applications, it is possible that a pending PCT application designating such a jurisdiction could be sufficient for a CL to be issued even if said PCT application has not yet entered the national/regional phase based on Art. 11(3) PCT which states that an “international application…shall have the effect of a regular national application in each designated State as of the international filing date”. This effect is explicitly called out under US patent law, but may not be elsewhere. Furthermore, PCT applications, and their national equivalents, are called out at 1.19 of the MPP-Pfizer license agreement which provides additional insight into how pending PCT applications may be considered from a licensing perspective.
Kickstarting the process of issuing CLs before the pending PCT application for nirmatrelvir enters the national/regional phase, which could be up to 31 months from the earliest priority date (i.e., Apr. 3, 2023 as outlined above), in the jurisdictions where this is possible, could facilitate access to a life-saving drug.
Conclusion
How readily drugs like Paxlovid can be accessed and utilised globally to stem the damaging effects of the COVID-19 pandemic is substantially dictated by supply and affordability of said drugs.
As of today, there are two generic manufacturers of Paxlovid in Bangladesh (Beximco and Eskayef, marketing the treatment as Bexovid and Paxovir, respectively) with emergency use authorizations to produce and sell generic versions of the drug for the local market. These companies have local production capacity for the drug and brought down the cost of a five-day treatment course from Pfizer’s $530 to less than $200. However, it is not yet clear that these versions can meet stringent regulatory requirements in other countries.
The wait for Paxlovid-related patents to be granted or refused could be years long, and this uncertainty could discourage generic competition unless governments invite that competition by licensing, or declaring a policy of licensing, and making government use of patents. Governments also should work proactively to clarify patent landscapes and ensure that generic competition will be available to help drive affordability and ample supply.
Annex 1: PCT Application Deeper Dive (WO/2021/250648)
Inventorship
Below is a table which lists each of the named inventors for the PCT application and outlines whether they were named as inventors on any of the four priority documents (P1-P4). It also details whether there appears to be an assignment from an inventor to Pfizer Inc. [US] based on what was disclosed in the “entitlement to claim priority” declaration form filed with the PCT application.
While we have not yet had sight of the assignment documents associated with each date in the below table, there appear to be a few instances—shaded in grey—in which an inventor has not been named on a priority application but has executed an assignment to Pfizer Inc. [US] for that application.
| P1: US 63/073982, 03.09.2020 | P2: US 63/143435, 29.01.2021 | P3: US 63/170158, 02.04.2021 | P4: US 63/194241, 28.05.2021 | |||||
| Named Inventor? | Date Assigned | Named Inventor? | Date Assigned | Named Inventor? | Date Assigned | Named Inventor? | Date Assigned | |
| OWEN, Dafydd Rhys |
Y | 09.02.2021 | Y | 09.02.2021 | Y | 08.04.2021 | Y | 01.06.2021 |
| PETTERSSON, Martin Youngjin |
Y | 03.02.2021 | Y | 03.03.2021 | Y | 06.04.2021 | Y | 01.06.2021 |
| REESE, Matthew Richard |
Y | 04.02.2021 | Y | 04.02.2021 | Y | 08.04.2021 | Y | 02.06.2021 |
| SAMMONS, Matthew Forrest |
Y | 05.02.2021 | Y | 05.02.2021 | Y | 07.04.2021 | Y | 01.06.2021 |
| TUTTLE, Jamison Bryce |
Y | 23.02.2021 | Y | 23.02.2021 | Y | 07.04.2021 | Y | 02.06.2021 |
| VERHOEST, Patrick Robert |
N | 27.02.2021 | Y | 08.02.2021 | Y | 08.04.2021 | Y | 02.06.2021 |
| WEI, Liuqing |
N | N/A | N | N/A | N | 15.06.2021 | N | 15.06.2021 |
| YANG, Qingyi |
Y | 26.02.2021 | Y | 26.02.2021 | Y | 07.04.2021 | Y | 01.06.2021 |
| YANG, Xiaojing |
N | N/A | N | N/A | Y | 12.04.2021 | Y | 02.06.2021 |
It would seem, then, that not all inventors were correctly listed on each priority application; however, this should be easily corrected by Pfizer without resulting in any CRISPR-like priority issues given that all the inventors named in the priority applications, plus Liuqing Wei, were named on the PCT application, and they all appear to have assigned their rights in the invention to Pfizer prior to the PCT application’s filing date (Aug. 6, 2021).
PCT Application Documents
Looking through each of the priority documents in chronological order, and finally the PCT application, it would seem that the way the “invention” has been defined and evolved coincided with the synthesis and testing of new compounds.
P1 is the first priority application that Pfizer filed in this family, and it framed how the invention went on to be defined in the subsequent priority applications and the resultant PCT application.
While nirmatrelvir was disclosed in P1 (see Example 13), it is not apparent that the compound was definitively thought of as the lead candidate at the time of its filing given that the claims do not narrow specifically around its structure and there is not a claim directed to nirmatrelvir only.
The features of the 74 compounds exemplified in P1 are distilled by Pfizer into the below Markush, Formula I, and there are fallbacks to preferred substituents in the claims and the description. It is interesting to see the PCT application related to “PF-00835231,” Pfizer’s antiviral drug candidate against SARS-CoV-1, referred to in P1 for the purpose of illustrating a synthetic route to obtain compounds of Formula I (see WO/2005/113580 – there appear to be no pending patent applications or granted patents in this family).
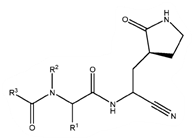 |
| Formula I |
P1 discloses compounds of Formula I and its formulations in various dosage forms, in combination with other agents (ritonavir is mentioned in the description, but not claimed), in methods of treating coronavirus infections (especially COVID-19), and so forth. Synthetic routes to the exemplified compounds are described as well as their biological activity against SARS-CoV-2.
Given that nirmatrelvir is disclosed in P1 and within the defined scope of its claims, it seems likely that claims to nirmatrelvir in the PCT and its related applications will be entitled to the priority date of P1 (i.e., Sep. 3, 2020). Priority dates are important as they determine the date from which the novelty and inventive step of claimed subject-matter should be assessedi.
P2 is an evolution of P1, and it appears as though the differences between the two applications were by addition. Ten new examples were added (Examples 75-84) which appear to have led Pfizer to introduce a new Markush, Formula I’ below, to cover these compounds (the notable difference between Formula I and Formula I’ being that oxo or hydroxy substituents, defined by (R)p, may be present on the 5-membered terminal ring of the latter).
 |
| Formula I’ |
It is more apparent from P2 that Pfizer was backing nirmatrelvir as the lead candidate at the time of its filing given that claims were introduced which single out nirmatrelvir (e.g., claims 52-60), including some which define preferred dose ranges. Additional synthetic routes for producing nirmatrelvir (see Example 13) were also introduced, along with information regarding predicted pharmacokinetic parameters.
Additional compounds also were listed for potential combination with compounds of Formula I’, and combinations with ritonavir were claimed (although ritonavir was not called out as a particularly preferred combination).
P3 is an evolution of P2, and it appears as though the differences between the two applications were by addition. Fourteen new examples were added (Examples 85-98), however these do not appear to be the reason behind Pfizer introducing a further Markush, Formula I’’ below (the notable difference between Formula I’ and Formula I’’ being that terminal ring of the latter may be a 4- to 8-membered ring).
 |
| Formula I’’ |
There are no examples in P3 where the terminal ring is not 5-membered, and changes to Formula I’ to arrive at Formula I’’ therefore seems to be somewhat speculative.
Further definitions were added to the description for terms such as “patient”, “pharmaceutically acceptable”, “therapeutically effective amount”, and so forth, as well as information regarding new variants of SARS-CoV-2. Further additional agents for combination with compounds of Formula I’’ were listed.
With regards to nirmatrelvir, further alternate synthetic routes were added (see Example 13) as well as recrystallization and X-ray crystallographic information; figures relating to structural details of nirmatrelvir and intermediates also were added. Ritonavir as a preferred combination agent (particularly with nirmatrelvir) also was specifically called out in the description and the claims, along with preferred dosage regimens.
P4 is an evolution of P3, and it appears as though the differences between the two applications were by addition. The additions mainly revolved around dosage regimens and spectral characterisations of the compounds.
PCT: WO/2021/250648, 06.08.2021
When turning to the contents of the PCT application (WO/2021/250648) itself, what is described and claimed in the application appears to be a refinement of the subject-matter disclosed in the four associated priority documents (P1-P4), with definitions and claims more succinctly formulated.
As mentioned in PCT Application Overview section, there are several Markush claims which broadly cover nirmatrelvir, and more narrowly defined claims which center around nirmatrelvir more specifically. It is interesting to note that a further compound, illustrated to the right,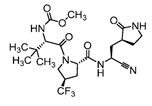 is also focused on specifically in the claims (see also Example 95, which was added in P3) in addition to nirmatrelvir; this indicates that Pfizer may be looking into its clinical important.
is also focused on specifically in the claims (see also Example 95, which was added in P3) in addition to nirmatrelvir; this indicates that Pfizer may be looking into its clinical important.
Pfizer filed their PCT application with the International Bureau and selected the European Patent Office to act as the ISA, meaning that a European patent examiner was the one to review the patentability of the application.
Based on the documents cited in the ISA’s search report, outlined below, the examiner determined that while the subject-matter of all the claims are novel, an inventive step could only be acknowledged for claims 2-16.
| Author or Applicant | Title | Relevant claims | Category | |
| D1 | Yangyang Zhai, et al. | Cyanohydrin as an Anchoring Group for Potent and Selective Inhibitors of Enterovirus 71 3C Protease | 1, 17-30 | Y – “document of particular relevance; the claimed invention cannot be considered to involve an inventive step when the document is combined with one or more other such documents, such combination being obvious to a person skilled in the art” |
| D2 | Yaxin Wang, et al. | Inhibition of enterovirus 71 replication by an α-hydroxy-nitrile derivative NK-1.9k | 1, 17-30 | Y – “document of particular relevance; the claimed invention cannot be considered to involve an inventive step when the document is combined with one or more other such documents, such combination being obvious to a person skilled in the art” |
| D3 | Bethany Halford | To conquer COVID-19, create the perfect pill | 1-30 | X – “document of particular relevance; the claimed invention cannot be considered novel or cannot be considered to involve an inventive step when the document is taken alone”
P – “document published prior to the international filing date but later than the priority date claimed” |
| D4 | Pfizer Inc. [US] | WO/2005/113580 – Anticoronviral compounds and compositions, their pharmaceutical uses and materials for their synthesis | 1, 17-30 | Y – “document of particular relevance; the claimed invention cannot be considered to involve an inventive step when the document is combined with one or more other such documents, such combination being obvious to a person skilled in the art” |
D4, which is Pfizer’s own prior art document relating to its earlier antiviral drug candidate against SARS-CoV-1 (“PF-00835231”), is cited by the examiner as the closest prior art. In essence, the examiner argues that Formula I’’ of claim 1 is too broadly defined and does not have enough differentiating features when compared to the compounds of D4 alone or in combination with the teachings of D1 or D2.
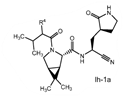
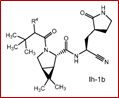
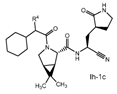
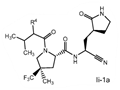
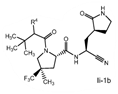
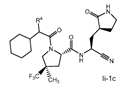
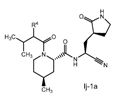
Claims 2-16 are, however, deemed to involve an inventive step by virtue of the presence of “the pyrrolidine or piperidine ring formed by R1 and R2 taken together with the nitrogen and the carbon atoms to which they are attached.” Claim 2 is defined below, with the Markush structure most closely related to nirmatrelvir shown surrounded by a red box.
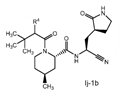
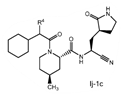
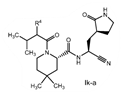
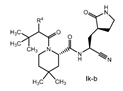
| 2. The compound of claim 1 selected from the group consisting of | |||
 , , |
 , , |
 , , |
 , , |
 , , |
 , , |
 , , |
 , , |
 , , |
 , , |
 , , |
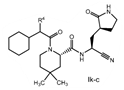 ; ; |
| wherein R4 is selected from the group consisting of (C1-C6 alkyl)amino optionally substituted with one to five fluoro, C1-C6 alkyl-C(O)NH- optionally substituted with one to five fluoro, and C1-C6 alkyl-S(O)2NH- optionally substituted with one to five fluoro; or a solvate or hydrate thereof, or a pharmaceutically acceptable salt of said compound, solvate or hydrate. | |||
The examiner also has identified a piece of prior art, D3, which will only become relevant should the claims not be entitled to the priority dates from before May 10, 2021 (i.e., the priority dates for P1-P3). While a full analysis of the priority date that each claim is entitled to is beyond the scope of this report, based on the analysis of the prior art documents outlined above, it does not appear that Pfizer will ultimately face issues on this front (for example, claim 2 of the PCT application finds basis in claims 11-12 of P1).
Acknowledgments
This report was written by Benjamin Wild. It was edited by Burcu Kilic, research director of the Access to Medicines Program, Peter Maybarduk, director of the Access to Medicines Program, and Brook Baker, senior adviser in Health Gap.
Endnotes
[i] By utilizing the “priority application” mechanism available in jurisdictions which are party to the Paris Convention, applications which validly claim priority are shielded from prior art which is disclosed after the earliest priority date when a claimed invention’s novelty and/or inventive step is assessed despite the fact that the national/regional application may not have been filed until after the prior art disclosure.
[ii] The time between filing the PCT application and filing national/regional applications therefrom is called the “international phase”.
[iii] The published document details a list of national and regional patent offices into which applications can be filed from the PCT application; at present, there are 153 contracting states. While the jurisdictions of interest to the applicant remain unclear to third parties at this point, publication does provide insight into the inventions for which protection is being sought.
[iv] Shortly after filing nationally/regionally, the separate applications will be republished by the relevant national/regional patent offices. While republication before national patent offices provides a clearer picture to third parties as to where the applicant intends to pursue patent protection for their invention, republication before regional patent offices (African Regional Intellectual Property Organization [ARIPO], Eurasian Patent Organization [EAPO], European Patent Organisation [EPO], and African Intellectual Property Organization [OAPI]) still leaves the door open to ambiguity as it is not possible to determine in which jurisdictions the applicant intends to make the patents effective until after grant.
[v] For an application to be granted, the invention being claimed must meet the patentability requirements of the patent office it is being prosecuted before; two key patentability hurdles that are common to most patent offices are that the claimed invention must be novel (i.e., its features must not have been disclosed in combination prior to the earliest priority date validly claimed) and possess an inventive step (i.e., it must not have been obvious for a person skilled in the art of the invention’s technical field to combine the claimed features to achieve a particular technical effect when starting from the prior art), although the standards a given patent office applies for these tests may differ from one to another. It is usual for claim amendments to be required during prosecution.
[vi] Some jurisdictions require annuities to be paid to patent offices prior to and post grant, and some only post. Once an application has been granted, it is possible for third parties to monitor whether an application remains in force by checking whether annuities are being paid, although not all patent offices provide up-to-date and readily accessible records to the public.
[vii] National jurisdictions where it is indicated that applications have been filed: Argentina, Aruba, Australia, Bangladesh, Bolivia, Brazil, Chile, Colombia, Costa Rica, Dominican Republic, Georgia, Guatemala, Honduras, India, Israel, Kosovo, Lebanon, Mexico, Mongolia, New Zealand, Nicaragua, Nigeria, Pakistan, Panama, Paraguay, Philippines, Russian Federation, Singapore, Taiwan, Ukraine, United Arab Emirates, United States of America, Uruguay, Venezuela (Bolivarian Republic of).
Regional jurisdictions where it is indicated that applications have been filed: ARIPO (Botswana, Eswatini, Gambia, Ghana, Kenya, Lesotho, Liberia, Malawi, Mozambique, Namibia, Rwanda, São Tomé and Principe, Sierra Leone, Sudan, Tanzania, Uganda, Zambia, Zimbabwe), EAPO (Armenia, Azerbaijan, Belarus, Kazakhstan, Kyrgyz Republic, Tajikistan, Turkmenistan, Russian Federation).
[viii] National jurisdictions where it is indicated that applications have been filed: Canada, Cuba, Peru, Republic of Korea, Saudi Arabia.
Regional jurisdictions where it is indicated that applications have been filed: EPO (Albania, Austria, Belgium, Bosnia and Herzegovina [extension state requested], Bulgaria, Cambodia [validation state not yet requested], Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Italy, Latvia, Liechtenstein, Lithuania, Luxembourg, Malta, Moldova [validation state requested], Monaco, Montenegro [extension state requested], Morocco [validation state requested], Netherlands, North Macedonia, Norway, Poland, Portugal, Romania, San Marino, Serbia, Slovak Republic, Slovenia, Spain, Sweden, Switzerland, Tunisia [validation state requested], Turkey, United Kingdom).
[ix] National jurisdictions where it is indicated that applications will be filed: Algeria, Bahamas, Bahrain, Barbados, China, Ecuador, Egypt, El Salvador, Hong Kong [registration via Chinese, European, or UK patent application], Indonesia, Japan, Kuwait, Macao [registration via Chinese patent application], Malaysia, Oman, Qatar, South Africa, Sri Lanka, Thailand, Trinidad and Tobago, Uzbekistan, Vietnam.
Regional jurisdictions where it is indicated that applications will be filed: OAPI (Benin, Burkina Faso, Cameroon, Central African Republic, Chad, Comoros, Congo, Côte d’Ivoire, Equatorial Guinea, Gabon, Guinea, Guinea-Bissau, Mali, Mauritania, Niger, Senegal, Togo).
[x] National jurisdictions where applications could additionally be filed via the pending PCT: Angola, Antigua and Barbuda, Belize, Brunei Darussalam, Democratic People’s Republic of Korea, Djibouti, Dominica, Grenada, Iran (Islamic Republic of), Jordan, Lao People’s Democratic Republic, Libya, Madagascar, Papua New Guinea, Saint Kitts and Nevis, Saint Lucia, Saint Vincent and the Grenadines, Samoa, Seychelles, Syrian Arab Republic.
Regional jurisdictions where applications could additionally be filed via the pending PCT: No further regional jurisdictions to select.
[xi] Argentina, Aruba, Bangladesh, Bolivia, Kosovo, Lebanon, Pakistan, Paraguay, Taiwan, Uruguay, Venezuela (Bolivarian Republic of).
[xii] Australia, Brazil, Canada, Chile, Colombia, Costa Rica, Cuba, Dominican Republic, Georgia, Guatemala, Honduras, India, Israel, Mexico, Mongolia, New Zealand, Nicaragua, Nigeria, Panama, Peru, Philippines, Republic of Korea, Russian Federation, Saudi Arabia, Singapore, Ukraine, United Arab Emirates, United States of America, ARIPO, EAPO, EPO.
[xiii] Search performed 01/01/2022.
[xiv] For additional background, an article summarizing other patents related to the field of SARS coronavirus protease inhibition can be found here.
[xv] Jurisdictions, sorted by World Bank income classification, which appear to have regulations which expressly1 or possibly2 allow for compulsory licenses/government use/Crown use of pending patent applications include (and may not be limited to): Low – Madagascar1*; Lower-middle – Algeria1*, Honduras1*, India1*, Nicaragua1*, Tunisia1*, Vietnam2*, Zambia1*; Upper-middle – Albania1, Costa Rica1, Dominican Republic1, Guatemala1*, Saint Lucia1, Turkey1; High – Australia1, Chile2, Cyprus1, Czech Republic2, France1, Greece2, Ireland1, Israel1, Latvia2, Malta1, Spain1, United Kingdom1.
*Mentioned in Exhibit C of the MPP-Pfizer license agreement (i.e., the “country list”).
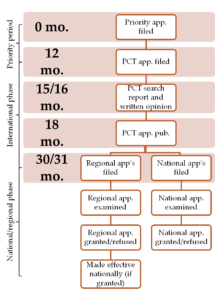 The patent application process typically starts with filing a priority application, usually in the applicant’s home country, which starts a 12-month clock for filing applications in other jurisdictions; before this clock has run out, national, regional, and/or international applications are filed claiming priority to the earlier application
The patent application process typically starts with filing a priority application, usually in the applicant’s home country, which starts a 12-month clock for filing applications in other jurisdictions; before this clock has run out, national, regional, and/or international applications are filed claiming priority to the earlier application