Letter Urging Pfizer to Address Paxlovid Inequality
UPDATE: Pfizer responded on January 31 (see PDF).
Albert Bourla
Chairman and Chief Executive Officer
Pfizer
235 E 42nd St
New York, NY 10017
January 24, 2022
Dear Mr. Bourla,
We are gravely concerned that inequalities in access to COVID-19 treatments will soon resemble, if not exceed, gaps in vaccine access seen around the world. We urge you to choose a better path for your oral antiviral treatment, Paxlovid, by significantly ramping up supply to developing countries.
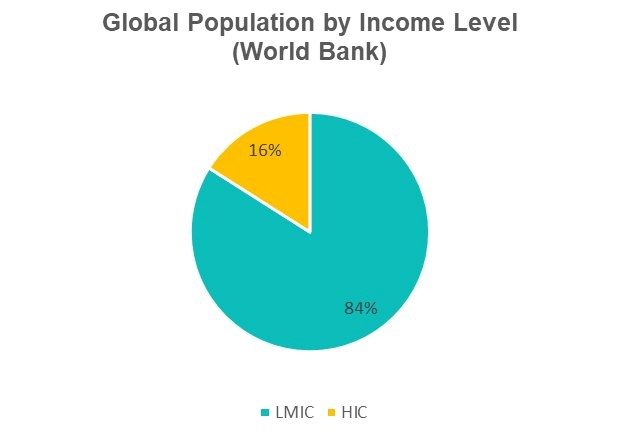
Last year, the head of the International Federation of Pharmaceutical Manufacturers and Associations admitted that the world “dismally failed” at the global COVID-19 vaccine rollout.[1] Pfizer, for its part, told investors that it was on track to deliver 60% percent of its vaccine doses to wealthy countries that we estimate represent just 16% of the world population.[2] The same mistake cannot be repeated for treatments.
Figure 1: Slide from Pfizer Investor Deck (Dec 17. 2021), and Public Citizen Estimate of Global Population by Income Level
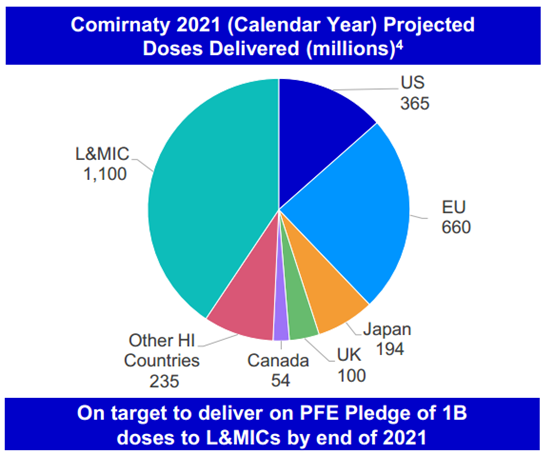
Pfizer expects to produce 120 million Paxlovid courses this year.[3] While the course of the pandemic remains uncertain, the need for Paxlovid may be significant. Paxlovid is currently authorized in the U.S. for people at high risk for severe COVID-19.[4] In 2020, Clark et al. estimated that more than half the global population at increased risk of severe COVID-19 lived in Africa, Asia and Latin America and the Caribbean.[5] But that is largely not where Paxlovid is being supplied. Instead, Pfizer has entered into Paxlovid supply agreements almost exclusively with countries based in North America and Europe, according to a tracker compiled by Knowledge Ecology International.[6] No African country has yet to purchase the treatment at all.
In 2021, Pfizer estimated that the addressable patient population would total 250 million in 2022.[7] 95 million patients would be in countries supplied by Medicine Patent Pool-licensed generics. However, according to a Pfizer investor document, those markets may “come on board” only in 2023, suggesting a delay of at least a year for robust supply from generics.[8] This is unacceptable.
Figure 2: Slide from Pfizer Investor Deck (Dec 17. 2021)
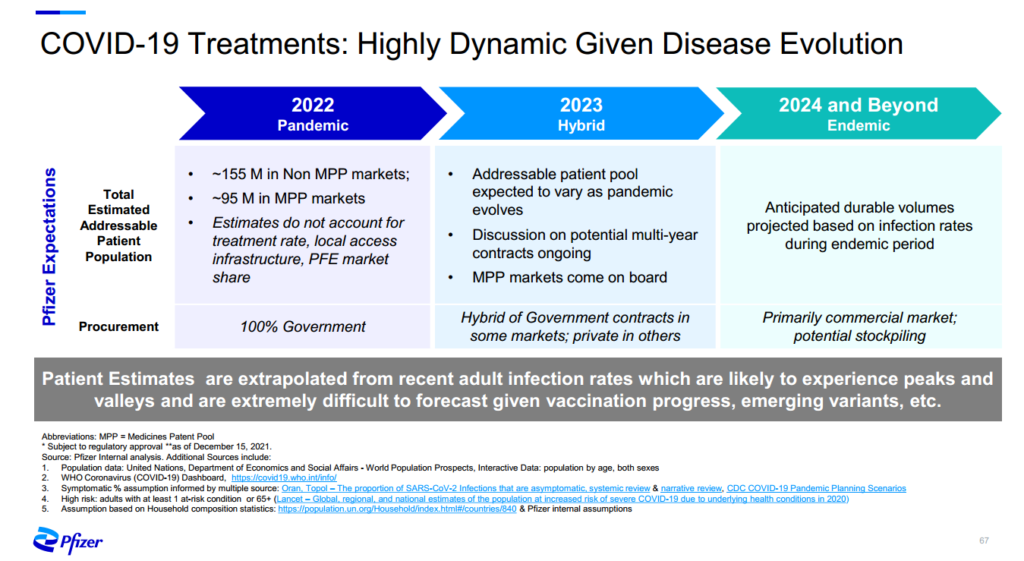
Pfizer can step up in three ways. First, it can set aside at least two-thirds of its 2022 supply for developing countries at a reasonable price, and make transparent its allocation criteria, pricing, and delivery schedules. Second, it can accelerate the entry of generic manufacturers licensed under the Medicines Patent Pool by providing deeper technical assistance and sharing regulatory information. Third, it can expand the scope of the Medicines Patent Pool license to allow the generic producers to supply more countries.
We urge Pfizer to help end the pandemic this year around the world—not just in a handful of rich countries.
Sincerely,
Peter Maybarduk
Director, Access to Medicines Program
Public Citizen
[1] https://www.telegraph.co.uk/global-health/science-and-disease/pharmaceutical-leaders-admit-dismally-failed-global-covid-vaccine/
[2] https://s28.q4cdn.com/781576035/files/doc_presentation/2021/12/17/COVID-Analyst-and-Investor-Call-deck_FINAL.pdf pg 29
[3] https://www.pfizer.com/news/press-release/press-release-detail/pfizer-provide-us-government-additional-10-million
[4] https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-first-oral-antiviral-treatment-covid-19
[5] 1.6 billion people who had one underlying condition or were above the age of 65 lived in Africa, Asia, and Latin America and the Caribbean. https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(20)30264-3/fulltext
[6] https://docs.google.com/spreadsheets/d/1fE1sB6VwrrqGTXReJb29IH_b-B6yeOhFRzsg0_D1GrQ/edit
[7] https://s28.q4cdn.com/781576035/files/doc_presentation/2021/12/17/COVID-Analyst-and-Investor-Call-deck_FINAL.pdf pg 67
[8] The Medicines Patent Pool aims to facilitate generic entry in the second half of 2022. https://s28.q4cdn.com/781576035/files/doc_presentation/2021/12/17/COVID-Analyst-and-Investor-Call-deck_FINAL.pdf pg 67