A False Choice Between Affordability and Innovation
Illinois Should Take Action to Lower Drug Costs
Illinois is currently considering legislation to address the burden imposed by high-priced prescription drugs. HB 1443 / SB 66 would establish a Prescription Drug Affordability Board (PDAB) that would analyze excessive drug costs and establish upper payment limits to help contain costs. Illinois’ proposed legislation would build on the success of the federal Medicare Drug Price Negotiation Program by adopting Medicare-negotiated prices as upper payment limits statewide.
The pharmaceutical industry often fights efforts to rein in prescription drug costs by claiming that attempts to make drugs more affordable will harm the ability to invest in research and development for new medicines. In reality, pharmaceutical companies do not set prices based on a drug’s research and development cost. Rather, shielded by monopoly protections, companies set prices based on what the market will bear and reap large profits in the process.
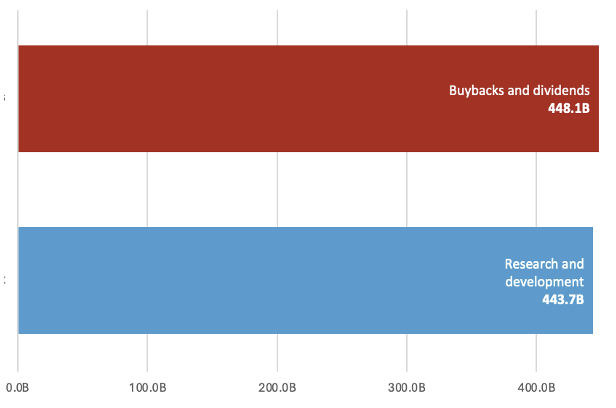
Pharmaceutical companies spend billions enriching shareholders through stock buybacks and dividends rather than investing in new treatments.[1] Over the past four years (2022 to 2025), the 15 publicly traded companies whose drugs were selected for the first and second round of Medicare negotiations collectively spent $4.4 billion more on stock buybacks and dividends than on research and development (Table 1 and Figure 1). For each individual year over the period, the companies collectively spent either more than or nearly as much on buybacks and dividends as they did on research and development. Notably, about half of the drugs (12/25) for which Medicare has already negotiated prices were among the top 25 highest-spend drugs for Illinois state health plans, which provides coverage for state employees and retirees. According to data obtained via a Freedom of Information Act (FOIA) request, these 12 drugs accounted for nearly $1 billion in spending for state health plans between fiscal years 2024 and 2025.[2]
Table 1. Spending on shareholder enrichment vs research and development among companies with drugs selected for the first and second round of Medicare price negotiations, 2022–2025
| Company | Medicare-selected drug(s), Rounds 1-2 | Drug(s) among the highest spend for Illinois state health plans FY24-25 | Buybacks & dividends | Research & development |
| Abbvie | Imbruvica, Linzess, Vraylar | 49.4B | 36.1B | |
| Amgen | Enbrel, Otezla | Y | 25.3B | 22.5B |
| Astellas | Xtandi | Y | 3.9B | 7.8B |
| AstraZeneca | Farxiga, Calquence | Y (Farxiga) | 18.4B | 48.5B |
| Bausch Health | Xifaxan | 0.0B | 2.4B | |
| Bayer | Xarelto | Y | 4.9B | 14.7B |
| Bristol Myers Squibb | Eliquis, Pomalyst | Y (Eliquis) | 32.4B | 39.9B |
| Eli Lilly | Jardiance | Y | 26.5B | 40.8B |
| GSK | Breo Ellipta, Trelegy Ellipta | Y (Trelegy Ellipta) | 15.4B | 32.6B |
| Johnson and Johnson | Stelara, Xarelto, Imbruvica | Y (Stelara, Xarelto) | 67.1B | 49.0B |
| Merck | Januvia, Janumet/XR | Y (Januvia) | 38.2B | 37.6B |
| Novartis | Entresto | Y | 67.1B | 41.8B |
| Novo Nordisk | Fiasp/Novolog, Ozempic/Wegovy/
Rybelsus |
Y (Ozempic/Wegovy) | 33.3B | 22.9B |
| Pfizer | Eliquis, Ibrance | Y (Eliquis) | 39.5B | 43.4B |
| Teva | Austedo/XR | 26.5B | 3.8B | |
| Total | 448.1B | 443.7B |
Public Citizen analysis of company SEC filings and analogous financial reports;[3] Illinois data obtained by Citizen Action/Illinois via a FOIA request to the Illinois Department of Central Management Service regarding prescription drug spending data for state health plans. FOIA data may not represent an exhaustive account of all state health plan drug costs. Values may not sum to the full total shown due to rounding.
Figure 1. Total spending on shareholder enrichment vs research and development among companies with drugs selected for the first and second round of Medicare price negotiations, 2022–2025
Similarly, in 2025, the 10 biggest drug corporations spent $84 billion on shareholder compensation and raked in $131 billion in profits, while spending $109 billion on research and development.
The companies for which Medicare has negotiated prices in the first two rounds of its negotiation program also spend more on financial payouts to shareholders than other companies. Based on a comparison of average financial payouts to shareholders (buybacks and dividends), these companies spend nearly three times as much on distributions to shareholders as other publicly traded companies in the S&P 500 (Figure 2).
Figure 2. Average distributions to shareholders among companies with drugs selected for the first and second round of Medicare price negotiations vs other S&P 500 index companies
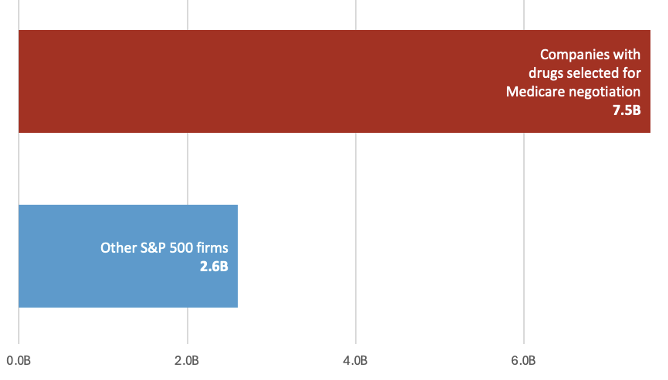
Public Citizen analysis of company SEC filings and analogous financial reports for Medicare-selected drugs (first and second round only), 2022-2025 and Twomey et al (2026) analysis of firms in the S&P 500 (excluding biopharmaceuticals, financial services, and real estate investment trusts), 2019-2023. This is intended to illustrate the wide gulf in average shareholder payouts between select pharmaceutical companies and other S&P 500 companies, but is not a one-to-one comparison as data were extracted over slightly different time periods.
Excessive drug pricing is even more egregious considering that taxpayer-funded contributions undergird virtually all new medicines. These contributions often focus on essential research on biological targets for drug action upon which drug development is based, de-risking critical parts of research and development. Drugs selected for the first round of Medicare negotiations collectively benefited from $12 billion in NIH funding (ranging from $4.5 million for insulin aspart and $6.48 billion for ustekinumab) for foundational research on drug targets and later-stage applied research prior to the drugs’ FDA approval.[4]
Pharmaceutical companies sell the same drugs at much lower prices in comparable countries and still make a profit at those prices. Bringing down drug prices, as the federal government has done for Medicare through its price negotiation program and as Illinois proposes in legislation to establish a PDAB, will rein in costs while still delivering lucrative revenues to drug corporations. Prices for the drugs selected for the first round of Medicare negotiations were on average over four times as expensive in the U.S. as in comparable countries, based on estimated U.S. net prices prior to negotiation.[5] Medicare-negotiated prices of those drugs remain higher than prices in other countries.
The pharmaceutical industry puts forward a false choice between making drugs more affordable and harming innovation. In reality, prescription drug corporations leverage government-granted monopolies to set prices to maximize revenues, and prices can be lowered significantly while preserving innovation. Medicare drug price negotiations have shown this at the federal level, with projections showing that the program will result in only 0.01% fewer new drugs over 30 years, while saving billions of dollars for patients and taxpayers over that same period. Illinois’ proposed PDAB legislation can build on Medicare negotiations for the 81 percent of its residents who are not on Medicare and can lower costs for drugs assessed to pose affordability challenges for which Medicare has not yet negotiated prices. Illinois should pass legislation to help ensure drug companies cannot put profits over the needs of everyday Americans struggling to afford their medications.
References
[1] Stock buybacks are a practice where a company repurchases shares, thereby inflating stock prices and enriching shareholders and executives often paid in stock. Dividends are another way publicly traded companies return cash to investors.
[2] Illinois data were obtained by Citizen Action/Illinois via a FOIA request to the Illinois Department of Central Management Service regarding prescription drug spending data for state health plans. Illinois state health plans spent $912.8 million between FY24 and FY25 on the twelve drugs that were both within the top 25 highest spend drugs, by ‘plan paid amount,’ across several state health plans over the two-year period and for which Medicare has negotiated prices over the first two rounds of the price negotiation program (Ozempic/Wegovy, Eliquis, Jardiance, Stelara, Xarelto, Enbrel, Xtandi, Farxiga, Entresto, Trelegy, Januvia, Otezla).
[3] For companies with multiple business segments, direct or general research and development expenses reported for their pharmaceutical segment was used. Boehringer Ingelheim was excluded from this analysis as it is a privately held company. All company figures represent full year financial reporting except for Astellas for FY2025 because it had not reported full year results at the time of writing.
[4] Zhou et al identified PubMed publications related to the drug target or drug and subsequently identified NIH grants associated with the publications. Basic research funding was totaled through the date of approval of a first-in-class product associated with that target (basic science funding for drugs with a common biological target is counted once).
[5] Wouters et al estimated 2021 net prices before negotiation and calculated the mean ratio of non-US prices to the estimated US net prices.