
A Plan for the People’s Vaccine
How the Biden Administration Can Supply the World
By Zain Rizvi and Peter Maybarduk
After Jonas Salk developed the polio vaccine, President Dwight Eisenhower paid tribute to the scientist in a Rose Garden ceremony. Salk’s work was “in the highest tradition of selfless and dedicated medical research,” said Eisenhower. “By helping scientists in other countries with technical information; by offering to them strains of seed virus and professional aid so that the production of vaccine can be started by them everywhere. . . Dr. Salk is a benefactor of mankind.”[1]
A safe and effective coronavirus vaccine could similarly prove a breakthrough for humanity—but only if it is available around the world quickly.
The moment calls for American leadership. Beneath some countries’ lofty rhetoric on cooperation, the situation appears bleak. The leading global vaccine initiative, COVAX, aims to supply only a fraction of humanity—two billion doses, likely enough for one billion people—by the end of 2021.[2] In May, a senior industry executive warned “When we think about what will be needed to address this pandemic, we are talking about orders of magnitude beyond what we as an industry are currently doing.”[3] In September, the world’s largest vaccine manufacturer noted a dose may not reach everyone until the end of 2024 at the earliest.[4]
A prolonged delay will be calamitous. The new coronavirus has already plunged the world into the worst health crisis in a century. The pandemic has battered economies, fueled inequality, and killed at least one million people. Years of delay may compound the challenges, with consequences for political stability and international relations. Closer to home, critical industries may remain at a standstill. The sheer magnitude of disparity—with a few million people in rich countries protected, with billions left behind—could ultimately resemble a global vaccine apartheid.
President-Elect Biden can choose a different path. The U.S. government has the knowledge, skills, and resources needed to help others find a way out of this pandemic. In this report, we trace how the Biden Administration can help the world make as much vaccine as possible, as quickly as possible. We show an alternative to scarcity: a dose of vaccine can be used by only one person, but all of humanity can benefit from the underlying knowledge about how to make the vaccine. A strategy focused on sharing the vaccine “recipe”, building manufacturing capacity, and mobilizing countries and corporations to meet the needs of the moment can accelerate the global response. It can also help restore U.S. leadership.
We proceed in four parts. First, we present the case for American action, describing the impact of the pandemic and gaps in the global response. Second, we explore the history of a little-known U.S. government program that helped dramatically scale-up flu vaccine manufacturing capacity around the world. Building on this model, we lay out three actions President-Elect Biden can take—mobilize, share, build—to quickly supply the world with a coronavirus vaccine. In doing so, we explain how the President has authority under existing laws, including the Defense Production Act, to implement this program. Finally, we conclude by presenting what a bold plan to supply vaccines could mean for America and for the world.[5]
Last month, more than 100 high-level leaders from public health, faith-based, racial justice, and labor organizations, including Public Citizen’s President, joined former members of Congress, economists and artists to sign a public letter calling on President-Elect Biden to make the COVID-19 vaccine a People’s Vaccine: a global public good, freely and fairly available to all, prioritizing those most in need here at home and around the world.[6] This report lays out a plan for how to get there.
Vaccine Apartheid
The coronavirus pandemic is the worst crisis since World War II, according to head of the United Nations.[7] Since the emergence of SARS-CoV-2 in Wuhan, 65 million cases have been reported.[8] More than 1.5 million people have died.[9] A U.N. report found that COVID-19 is “reversing decades of progress on poverty, healthcare and education.”[10] Lockdowns to contain the virus triggered the most severe economic downturn since the Great Depression.[11] The virus is now expected to push 100 million people into extreme poverty.[12] Life expectancy will fall.[13] Nearly twice as many people will face hunger.[14] At home, critical industries that support hundreds of thousands of American workers may not recover for years.[15] IMF economists predict that “the COVID-19 pandemic will likely increase inequality, trigger social unrest, and lower future output in the years to come.”[16]
A potential vaccine offers hope. But unequal access may only exacerbate the global challenges, leading to an uneven and unstable economic recovery and prolonged human suffering. The global effort to provide vaccines is important but insufficient. Rich countries have already bought the majority of existing vaccine supply.[17] The leading global initiative, COVAX, aims to supply 2 billion doses by the end of 2021—if everything goes right and all funding targets are met. So far, it has procured 700 million doses.[18] More than ten billion vaccine doses may ultimately be needed, with demand only increasing if the vaccines provide limited immunity and require booster shots.[19] A narrow focus on donations and distribution overshadows the possibility of rapidly expanding supply.
Figure 1: COVID-19 Vaccine Candidate Agreements[20]
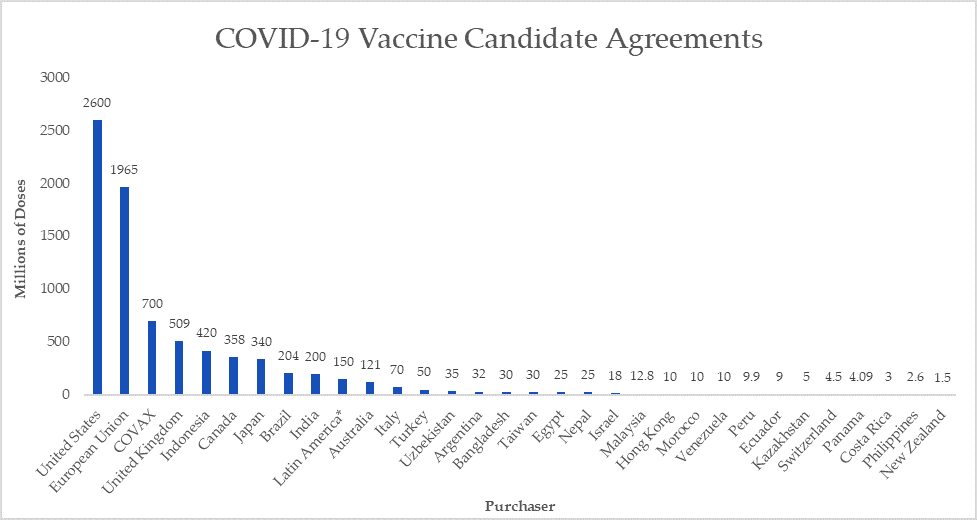
Some countries have pushed hard on vaccine diplomacy. President Xi Jinping told the World Health Assembly that a vaccine would be a “global public good,”[21] though he did not specify his meaning. Last month, the Chinese leader pledged to work with BRICS countries “to advance collective vaccine research and trials, set up plants [and] authorize production.”[22] Chinese manufacturers are rapidly scaling-up. In a UNICEF survey, Chinese manufacturers reported that they expect to double COVID vaccine production capacity by 2022.[23] By 2023, China may account for half of all global production.[24] However, critical questions remain unanswered regarding the safety, efficacy, and distribution of potential Chinese vaccines.
The Biden Administration can step in to meet the needs of the moment. The U.S. government has played an important role in scaling up vaccine supply in the past.
“Helping Countries Help Themselves”: BARDA’s Influenza Program
“Diseases do not respect borders so increasing the ability to make flu vaccine in any country helps every country reduce the spread of flu,” explained Robin Robinson, then director of the U.S. Biomedical Advanced Research and Development Authority, in 2010.[25] “While we continue to build vaccine manufacturing capacity in the United States, and develop new, faster technologies for producing vaccine domestically, we can increase pandemic preparedness worldwide by helping other countries take advantage of technology.”
Robinson was referring to BARDA’s international program, launched years earlier, to help developing countries build influenza vaccine manufacturing capacity. The spread of the deadly H5N1 influenza strain had raised alarm bells in 2004. After a global call for action at the World Health Assembly, the Bush Administration launched a new program.[26]
The impact was significant. In 2005, vaccine producers in the countries involved could collectively produce less than 1 million doses of pandemic influenza vaccine. In 2014, they could produce nearly 300 million doses. In 2018, they could produce over 500 million doses.[27]
Figure 2: BARDA Global Influenza Vaccine Partners (Blue/Yellow) (2016)[28]
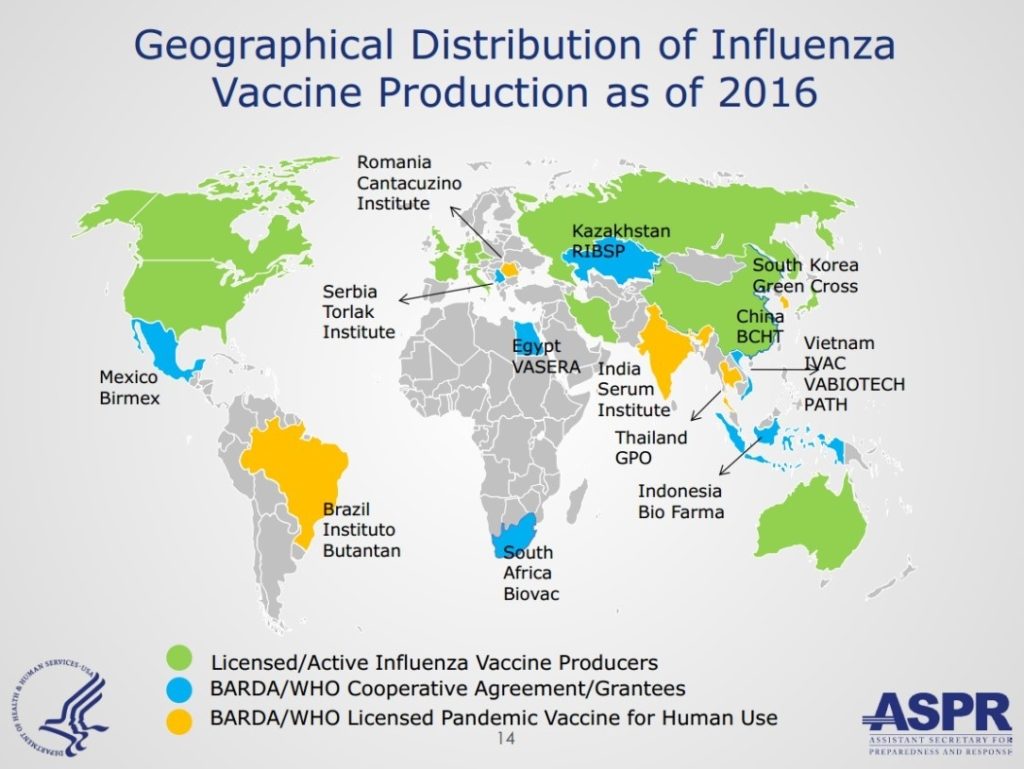
Behind the success lay a novel partnership with the World Health Organization (WHO), which was implementing a global influenza plan, and developing country manufacturers. Alongside the WHO, BARDA helped build facilities, train personnel, provide technical assistance, and transfer technology for scalable manufacturing.[29] WHO obtained an intellectual property license from a Russian institute on a vaccine strain so that manufacturers could more easily begin production, and created a vaccine technology hub.[30] BARDA and WHO supported 14 manufacturers in 13 countries.[31] While funding data is not publicly available, the U.S. government reported investing $72 million by 2013.[32] Every dollar invested by BARDA leveraged seventeen dollars of local investment.[33] Congress expressed strong support. In 2013, the Senate Committee on Appropriations pushed back against proposed funding cuts and noted that “The capability of developing countries to produce influenza vaccine within their borders is crucial to reducing the threat of a global pandemic.”[34]
In a sense, the BARDA program was merely a pilot for what is needed now: a U.S. government initiative to rapidly increase vaccine supply by sharing technology, building capacity, and mobilizing the global community. There are important differences. The leading coronavirus vaccine candidates rely on newer technology as compared to traditional egg-based approaches used in many influenza vaccines, raising the prospect of more complex technical processes and intellectual property claims. But the BARDA international program offers an important vision. “We are helping countries help themselves,” said Robin Robinson, BARDA’s former director. [35] Now, the U.S. government should rise to meet the needs of this moment, and inject American ambition into the global response.
The Plan
Mobilize
President-Elect Biden should launch a new program focused on supplying the world with a coronavirus vaccine. The program should aim to make as much vaccine as possible, as quickly as possible.
Getting there will require a global collaboration. No one corporation can supply the world, and fragmented licensing arrangements between corporations will not get us there quickly enough. President-Elect Biden should commit to sharing the know-how and intellectual property needed to produce a vaccine, so manufacturers around the world can quickly start ramping up production. He should also build additional manufacturing capacity.
A bold plan from the United States will energize the global response, building on nascent momentum to develop a sharing solution. 34 states have endorsed a WHO-led initiative to create a pool for sharing COVID-19 technology.[36] India and South Africa have a proposed a waiver on COVID-19 related intellectual property at the World Trade Organization.[37] This could help free up space for sharing technology around the world—a move the humanitarian organization Doctors Without Borders called a “gamechanger.”[38] President Emmanuel Macron recently noted that “sharing knowledge and know-how must also be at the heart of our [vaccine] strategy.”[39] A serious American program backing these efforts could galvanize others. Countries and corporations alike would benefit from a reciprocal approach to sharing scientific knowledge and technology.[40]
At home, President-Elect Biden can begin with a call to action. He can convene vaccine manufacturers and ask what technology they are willing to share to bring a rapid end to the pandemic. Industry executives, responding to the President on how they would meet a once-in-a-century crisis, may find it hard to resist the humanitarian imperative. The President can negotiate with the manufacturers, fairly compensating them for sharing technology and relevant intellectual property, taking into account the substantial public investment in developing the vaccines. If the corporations were unwilling to share, the President could use his existing legal authority to require sharing while providing fair compensation, as described in the section below.
Table 1: Direct Public Support of Leading Coronavirus Vaccine Candidates[41]
| Group | Operation Warp Speed Funding[42] | Other Support |
| Moderna/National Institutes of Health
(mRNA-1273) |
$901,894,979 (R&D)
$1,578,000,000 (Manufacturing) $2,479,894,979 (Total) |
· NIH helped invent the vaccine, claims joint ownership, and is running clinical trials.[43]
· Candidate uses NIH spike protein technology.[44] · Moderna has said 100% of the activities covered under its BARDA contract—ranging from clinical trials to FDA application fees—are taxpayer-funded.[45] |
| AstraZeneca/Oxford
(AZD1222) |
$1,600,000,000 (R&D and Manufacturing) | · Oxford invented the vaccine and is helping run clinical trials.
· NIH is also helping run and fund Phase III clinical trials.[46] · Coalition for Epidemic Preparedness Innovations provided $384 million.[47] · A publicly-funded facility is supporting manufacturing.[48] |
| Johnson & Johnson
(JNJ-78436735) |
$910,237,081 (R&D)[49]
$1,001,650,000 (Manufacturing) $1,911,887,081 (Total) |
· NIH is helping run and fund Phase III clinical trials.[50]
· Candidate uses NIH spike protein technology.[51] · A publicly-funded facility is supporting manufacturing.[52] |
| Novavax
(NVX-CoV2373) |
$1,600,434,523 (Manufacturing) | · Candidate uses NIH spike protein technology.[53]
· Coalition for Epidemic Preparedness Innovations provided $388 million.[54] · A publicly-funded facility is supporting manufacturing.[55] |
| Pfizer/BioNTech
(BNT162b2) |
$1,950,000,000 (Purchase commitment) | · Candidate uses NIH spike protein technology.[56]
· BioNTech received €100 million loan from European Investment Bank and €375 million in funding from German government.[57] · Operation Warp Speed helped with logistics, such as securing raw materials.[58] |
| Sanofi/GSK
(unknown) |
$2,072,275,336 (R&D and manufacturing) |
Building on the federal government’s earlier investments in coronavirus research, Operation Warp Speed has helped spur the development of promising vaccine candidates targeting the new coronavirus in record time—a remarkable scientific achievement.[59] If the vaccines prove safe and effective, the Biden Administration can help guide humanity out of this pandemic by sharing technology and building manufacturing capacity.
Share
Tapping all existing manufacturing capacity is critical to rapidly supplying the world with a vaccine. A process called “technology transfer” can help scale-out manufacturing across different production sites. The complexity of the technology and the capabilities of the recipient influence the speed of technology transfer, but publicly available information suggests transfer is possible within a few months.
Table 2: Examples of COVID-19 Technology Transfer[60]
| Group | Description | Estimated Timeline |
| AstraZeneca/Oxford Emergent Biosolutions | Corporations announced technology transfer agreement in June 2020.[61] Emergent began large-scale production in September.[62] | ~4 months |
| AstraZeneca/Oxford
Serum Institute of India |
Corporations announced agreement in June.[63] Serum Institute indicated it was close to large-scale production end of July.[64] By November, Serum Institute had produced 40 million doses.[65] | ~2 months to 5 months |
| Moderna/NIH
Lonza |
Technology transfer began June 2020. Lonza produced first batches in July 2020, likely for clinical trial doses.[66] Commercial scale production is expected by December.[67] | ~2 months to 7 months |
| McKinsey Estimate | “We believe [industry] can achieve best-in-class transfer times of eight to 11 months, 60 to 70 percent more quickly than usual, at scale. With regulatory flexibility that has already been announced, it may be possible in six.”[68] | ~6 months to 11 months |
During the pandemic, most manufacturers, driven by commercial priorities, have kept technology secret or limited transfer to a handful of producers. (One exception has been AstraZeneca, which was pushed by its academic partner Oxford to share know-how with producers around the world, helping bolster global supply. But even AstraZeneca’s sharing is conditioned on meeting secret requirements.[69])
Limited sharing means that existing vaccine manufacturing capacity may sit idle even as the world faces vaccine shortages. Consider, for example, that 52 manufacturers told the global COVAX initiative they had capacity to supply the “drug substance” of a vaccine.[70] Of those, COVAX reached out to 20 groups.[71] Only 7 deals have been signed.[72] This is consistent with an earlier survey conducted by the Developing Countries’ Vaccine Manufacturers’ Network, which found that half their members were engaged in the pandemic response.[73]
The U.S. government is uniquely positioned to lead the sharing effort. It has both the scientific and political muscle. On a technical level, the government can build on its experience facilitating technology transfer domestically through Operation Warp Speed and globally through the BARDA international program.[74] Given its deep scientific expertise, the government can also develop and disseminate best-practices, provide rapid-response technical assistance,[75] and work with the WHO’s COVID-19 technology access pool to reciprocally share knowledge.[76]
In addition, the U.S. government has significant political leverage to ask corporations to meet the humanitarian imperative and share information. Corporations typically maintain thousand page dossiers that describe manufacturing data, processes and protocols (i.e., the recipe).[77] If negotiations fail, the government has legal leverage to require corporations to act in the public interest. First, the government typically maintains rights in the intellectual property and data it has helped develop—through funding or the intellectual contributions of federal scientists.[78] BARDA alone has provided $14 billion to advance coronavirus research, development and manufacturing, and the NIH jointly developed the NIH-Moderna vaccine.[79] While the Trump Administration has weakened some rights, others can help facilitate sharing.[80] The Obama Administration, for example, was the first to share patents related to an HIV medicine with the global community through the Medicines Patent Pool.[81] Second, the government has broad authority under existing law to facilitate sharing of intellectual property and know-how. The Defense Production Act, for example, contains a broad information disclosure obligation. Under Section 1498, the government can use a patent in exchange for reasonable compensation.[82] If needed, these provisions can be used creatively to make vaccines global public goods. Additional legislation could strengthen this mandate.
Table 3: Legal Authorities Related to Intellectual Property and Information Disclosure
| Authority | Provision | Use | Considerations |
| Worldwide Licenses
Bayh-Dole Act, analogous statutes, funding contracts[83] |
E.g., Bayh-Dole: “The Federal agency shall have a nonexclusive, nontransferrable, irrevocable, paid-up license to practice or have practiced for or on behalf of the United States any subject invention throughout the world.” | Give permission to global partners to use government-funded or government-owned medical inventions and data. | – Bayh-Dole contracts have standard set of rights.
– Scope of rights in other kinds of funding contracts vary.[84] – Government has vast authority for inventions it owns. |
| Information Disclosure Defense Production Act
50 U.S.C. § 4555 |
“The President shall be entitled. . . by regulation, subpoena, or otherwise, to
obtain such information from. . . any person as may be necessary or appropriate, in his discretion, to the enforcement or the administration of this Act. . . Information obtained under this section which the President deems confidential or with reference to which a request for confidential treatment is made by the person furnishing such information shall not be published or disclosed unless the President determines that the withholding thereof is contrary to the interest of the national defense.” |
Share technical information with global partners. | – National defense includes “military or critical infrastructure assistance to any foreign nation.”
– Recently used to assess industrial capabilities. – President can issue regulations to change scope and purpose of information disclosures. |
| Government Patent Use
28 U.S.C. § 1498 |
“Whenever an invention described in and covered by a patent of the United States is used or manufactured by or for the United States without license of the owner thereof or lawful right to use or manufacture the same, the owner’s remedy shall be by action against the United States in the United States Court of Federal Claims for the recovery of his reasonable and entire compensation for such use and manufacture.” | Clear patent barriers domestically, authorize multiple suppliers, and send signal to other countries to use similar authorities. | – Used regularly by federal government to procure everything from military equipment to electronic passports.[85]
– Government use cannot be enjoined. |
Build
Building more manufacturing capacity is also critical to quickly supplying the world with a vaccine. In May, a senior pharmaceutical executive testified “the industry will need to approximately double its current manufacturing capacity” to meet global demand for coronavirus vaccines.[86] The pharmaceutical industry was “already operating close to full capacity.”[87]
It remains unclear how much progress has been made since then. Capacity data is opaque and sometimes unreliable. A leader of the global COVAX initiative recently indicated they have “major gaps” when it comes to understanding the capacity of developing country manufacturers.[88] Estimates on production can vary by tenfold depending on yield and dosing requirements.[89]
Using its global reach, the U.S. government should identify capacity gaps, retrofit existing facilities, and build new factories. Following BARDA’s international program and Operation Warp Speed, the government should explore expanding capacity in developing countries and supplementing domestic capacity. Warp Speed notably augmented and scaled-up 23 manufacturing facilities within six months.[90] It also supported “equipment fitting, staff hiring and training, raw-material sourcing [and] technology transfer and validation.”[91] While building new factories may take time, Operation Warp Speed has shown how focused government action can quickly overcome perceived technical limitations. Even private corporations have learned to stand up new facilities during the pandemic within eight months.[92] Developing a sustainable source of vaccine production for this pandemic and the next should be considered a priority.
Building manufacturing capacity can also benefit from the U.S. military’s logistics capabilities. During the Ebola outbreak in West Africa in 2014, for example, the military set up field hospitals, operated medical labs, trained healthcare workers and ensured personal protective equipment supply.[93]
President-Elect Biden can rely on existing authority, including the Defense Production Act (DPA) and the Pandemic and All-Hazards Preparedness Act (PAHPA), to jumpstart the response. Title I of DPA allows the President to require businesses to prioritize contracts that promote the national defense.[94] This has been used to ensure a rapid supply of materials needed to produce vaccines for Operation Warp Speed.[95] Title III allows the President to provide economic incentives, and procure and install equipment in private facilities. PAHPA allows the government to build pandemic vaccine factories.[96] This has been used to setup three public-private manufacturing facilities that are now developing coronavirus vaccines.[97]
Conclusion
The coronavirus crisis will have a profound impact on the world. How long its scars last depend, in part, on how President-Elect Biden chooses to respond. By mobilizing the global community, sharing the vaccine recipe and building manufacturing capacity, President-Elect Biden can save lives, propel economies, and restore American leadership.
In his 2003 State of the Union address, President Bush proposed an Emergency Plan for AIDS Relief. “Seldom has history offered a greater opportunity to do so much for so many,” said Bush.[98] PEPFAR was made possible by the introduction of low-cost medicines by generic suppliers around the world. It has since provided treatment to 17 million people, transforming the global response to HIV/AIDS.[99] A coronavirus vaccine program can do even more for America and for the world. President-Elect Biden should help bring America back.
[1] Citations by President Eisenhower to Dr. Jonas E. Salk and the National Foundation for Infantile Paralysis, The White House (Apr. 22, 1955), https://tinyurl.com/y6kaapxh
[2] Seth Berkley, COVAX Explained, Gavi (Sep. 3, 2020), https://www.gavi.org/vaccineswork/covax-explained
[3] Testimony of Dr. Julie L. Gerberding, U.S. House of Representatives (Jul. 21, 2020), https://tinyurl.com/y6xurl8s
[4] “Not enough Covid vaccine for all until 2024, says biggest producer,” Financial Times (Sept. 13, 2020), https://tinyurl.com/y4vtwb8l
[5] Public Citizen is part of The Peoples’ Vaccine Alliance, a coalition of global and national organizations and activists united under a common aim of campaigning for a ‘People’s Vaccine’. The call for a People’s Vaccine is backed by past and present world leaders, health experts, faith leaders and economists. For more information visit: https://peoplesvaccine.org
[6] An Open Letter to President-Elect Biden, (Nov. 24 2020) https://oxfamus.medium.com/an-open-letter-to-president-elect-biden-acfb813549ab
[7] Edith Lederer, UN chief says COVID-19 is worst crisis since World War II, Associated Press (Mar. 31, 2020), https://tinyurl.com/y58c7xm2
[8] COVID-19 Dashboard, Johns Hopkins University, https://coronavirus.jhu.edu/map.html
[9] Id.
[10] UN report finds COVID-19 is reversing decades of progress on poverty, healthcare and education, United Nations (Jul. 7, 2020), https://tinyurl.com/y2deyyn2
[11] The Great Lockdown: Worst Economic Downturn Since the Great Depression, IMF (Apr. 14, 2020), https://tinyurl.com/yxxebmlp
[12] Updated estimates of the impact of COVID-19 on global poverty: The effect of new data, World Bank (Oct. 7, 2020), https://tinyurl.com/yxorfvbs
[13] Mark Lowcock, Foreword by the Emergency Relief Coordinator, UN OCHA, https://tinyurl.com/yxk72xrx
[14] Id.
[15] Patrick McGeehan, Tourism, Engine for N.Y.C. Economy, May Not Fully Recover Until 2025 (Nov. 16 2020), https://www.nytimes.com/2020/11/16/nyregion/nyc-tourism-covid.html
[16] Tahsin Saadi Sedik and Rui Xu, A Vicious Cycle: How Pandemics Lead to Economic Despair and Social Unrest, IMF (Oct. 2020), https://tinyurl.com/y6f8kp3b (“We established that past pandemics, even though much smaller in scale than COVID-19, have significantly contributed to social unrest through their impact on economic growth and inequality. Specifically, we provide evidence that pandemics tend to depress economic growth and increase inequality, and both lower growth and greater inequality are important drivers of social unrest. Furthermore, social unrest, in turn, is associated with output loss and with higher inequality, suggesting a vicious cycle. Our results would imply a heightened risk of social unrest post COVID-19 unless swift and bold policies are implemented to protect the most vulnerable group in the society.”)
[17] Ashe Mullard, How COVID vaccines are being divvied up around the world,” Nature (Nov. 30, 2020), https://tinyurl.com/y52pv3h5
[18] Id.
[19] Lea Frederiksen et al., The Long Road Toward COVID-19 Herd Immunity: Vaccine Platform Technologies and Mass Immunization Strategies, Front. in Immunology (July 21, 2020), https://tinyurl.com/y34wmsvx (“Based on this estimate, ~5.3 billion vaccine doses are required for a single-dose vaccine, or possibly 12–16 billion in case of a multi-dose vaccine.”)
[20] COVID-19 Vaccine Market Dashboard, UNICEF, https://www.unicef.org/supply/covid-19-vaccine-market-dashboard (Latin America figure excludes Brazil. Data retrieved from UNICEF database on December 7 2020.)
[21] Sarah Wheaton, Chinese vaccine would be ‘global public good,’ Xi says, Politico (May 18, 2020), https://tinyurl.com/yyp59k6m
[22] Full Text: Remarks by Chinese President Xi Jinping at 12th BRICS Summit, Xinhua Net (Nov. 17, 2020), https://tinyurl.com/y6oos47r
[23] Expression of Interest on supply of COVID-19 vaccines on behalf of the COVAX Facility, UNICEF (Aug. 31, 2020), https://tinyurl.com/y2skm3fu
[24] Id.
[25] BARDA Grants Help Build Global Flu Vaccine Manufacturing Capacity, Business Wire (Sep. 30, 2010), https://tinyurl.com/y2hcl72r
[26] Michael L. Perdue and Rick A. Bright, United States of America Department of Health and Human Services support for advancing influenza vaccine manufacturing in the developing world, Vaccine 29 (Jul. 1, 2011): A48-A50, https://tinyurl.com/y3sbjjmz In 2005, the World Health Assembly passed a resolution calling for increased developing country vaccine capacity. The U.S. Homeland Security Council then echoed this call in its National Strategy for Pandemic Influenza Implementation Plan, urging international cooperation to develop vaccine manufacturing capacity.
[27] Changing the Landscape for Global Pandemic Influenza Vaccine Manufacturing, ASPR (Feb. 13, 2014), https://tinyurl.com/y4nn23dk. (BARDA projected they would be able to produce up to 500 million doses by end of 2016.) Global Partnerships and International Preparedness: Challenges and Innovative Partnering, ASPR (Oct. 2016), https://tinyurl.com/y23ub6y3 .The head of the program, Rick Bright, noted in 2018 that “Pandemic vaccine manufacturing capacity in developing countries has expanded to over 500 million doses from this program.”
[28] Id.
[29] International Influenza Vaccine Manufacturing Capacity Building Program, Medical Countermeasures, https://tinyurl.com/yyr4cs79
[30] Friede, Martin, et al, WHO initiative to increase global and equitable access to influenza vaccine in the event of a pandemic: supporting developing country production capacity through technology transfer, Vaccine 29 (Jul. 1, 2011): A2-A7, https://tinyurl.com/yy2l8x5j
[31] Id.
[32] Julie Schafer, BARDA International Influenza Vaccine Manufacturing Capacity Building Program, ASPR, https://tinyurl.com/y2hwpxjp
[33] International Influenza Vaccine Manufacturing Capacity Building Program, Medical Countermeasures, https://tinyurl.com/yyr4cs79
[34] Departments of Labor, Health and Human Services, and Education, and Relateed Agencies Appropriation Bill, U.S. Senate (2013), https://tinyurl.com/y2cbbth6
[35] Jason deBruyn, NCSU’s B TEC moves to front lines in bio education, Business Journals (Apr. 15, 2011), https://tinyurl.com/y39kg7je
[36] Endorsements of the Solidarity Call to Action, WHO, https://tinyurl.com/y2uq426z
[37] India and South Africa proposal for WTO waiver from IP protections for COVID-19-related medical technologies, Doctors Without Borders (Nov. 18, 2020), https://tinyurl.com/y6o3fcj9
[38] Doctors Without Borders, 5 reasons a new proposal by India and South Africa could be a gamechanger in the COVID-19 response, (Oct. 11 2020), https://tinyurl.com/y4hj5xoj
[39] Intervention du Président Emmanuel Macron sur la riposte sanitaire et la préparation aux futures pandémies au sommet du G20, Elysee (Nov. 21, 2020), https://tinyurl.com/y27q8tmq
[40] For example, open science could help promote learning and accelerate candidate development. Consider the case of Pfizer and Moderna, which are developing two vaccines using very similar technology, but significantly different cold chain requirements. Sharing manufacturing processes, data and insights could have helped improve the candidates.
[41] Many of the candidates have also benefited from years of public investment in the underlying platform technology. See https://tinyurl.com/yyqr9qs3
[42] Contract amounts collected from the Medical Countermeasures website unless otherwise specified. COVID-19 Medical Countermeasure Portfolio, HHS, https://www.medicalcountermeasures.gov/app/barda/coronavirus/COVID19.aspx
[43] Bob Herman, The NIH claims joint ownership of Moderna’s coronavirus vaccine, Axios (Jun. 25, 2020), https://tinyurl.com/y9panleq. and “Promising Interim Results from Clinical Trial of NIH-Moderna COVID-19 Vaccine,” NIH (Nov. 16, 2020), https://tinyurl.com/y4tj7duz
[44]The NIH worked with scientists at the University of Texas to develop this technology. See https://tinyurl.com/yyegpfb6
[45] Bob Herman, Moderna skirts disclosures of coronavirus vaccine costs, Axios (Aug. 5, 2020), https://tinyurl.com/y3nkdks5
[46] “Phase 3 Clinical Testing in the US of AstraZeneca COVID-19 Vaccine Candidate Begins,” NIH, (Aug. 31, 2020), https://tinyurl.com/y2q5kfln
[47] Zain Rizvi, Covax’s Choices, Public Citizen (Nov. 16, 2020), https://www.citizen.org/article/covaxs-choices/
[48] Emergent BioSolutions Signs Agreement with AstraZeneca to Expand Manufacturing for COVID-19 Vaccine Candidate, Emergent BioSolutions (Jul. 27, 2020), https://tinyurl.com/y6su2tn2
[49] Johnson & Johnson and U.S. Department of Health & Human Services Expand Agreement to Support Next Phase of COVID-19 Vaccine Candidate Research and Development, Johnson & Johnson, (Nov. 14, 2020), https://tinyurl.com/y3u2mdjv
[50] Fourth large-scale COVID-19 vaccine trial begins in the United States, NIH, (Sep. 23, 2020), https://tinyurl.com/y22szdsd
[51] Zain Rizvi, Leading COVID-19 Vaccine Candidates Depend on NIH Technology, Public Citizen (Nov. 10, 2020), https://tinyurl.com/yyegpfb6
[52] Emergent BioSolutions Signs Five-Year Agreement for Large-Scale Drug Substance Manufacturing for Johnson & Johnson’s Lead COVID-19 Vaccine Candidate, Emergent BioSolutions (Jul. 6, 2020), https://tinyurl.com/y49qtb63
[53] Zain Rizvi, Leading COVID-19 Vaccine Candidates Depend on NIH Technology, Public Citizen (Nov. 10, 2020), https://tinyurl.com/yyegpfb6
[54] Zain Rizvi, Covax’s Choices, Public Citizen (Nov. 16, 2020), https://www.citizen.org/article/covaxs-choices/
[55] FEDS TAP BIO-MANUFACTURING CENTER IN COLLEGE STATION FOR “WARP SPEED” PRODUCTION OF COVID-19 VACCINES, Texas A&M University System (Jul. 27, 2020), https://tinyurl.com/y3yx4fwy
[56] Zain Rizvi, Leading COVID-19 Vaccine Candidates Depend on NIH Technology, Public Citizen (Nov. 10, 2020), https://tinyurl.com/yyegpfb6
[57] Investment Plan for Europe: European Investment Bank to provide BioNTech with up to €100 million in debt financing for COVID-19 vaccine development and manufacturing, BioNTech (Jun. 11, 2020), https://tinyurl.com/y68uycwv and BioNTech Gets $445 Million in German Funding for Vaccine, Bloomberg (Sep. 15, 2020), https://tinyurl.com/y6xx4feh
[58] US vaccine tsar calls on White House to allow contact with Biden, Financial Times (Nov. 13, 2020), https://tinyurl.com/y3w63j7z
[59] Zain Rizvi, Blind Spot, Public Citizen (Feb. 19, 2020), https://www.citizen.org/article/blind-spot/ (noting NIH alone has spent nearly $700 million on coronavirus research since the SARS epidemic). Zain Rizvi, Leading COVID-19 Vaccine Candidates Depend on NIH Technology, Public Citizen (Nov. 10, 2020), https://tinyurl.com/yyegpfb6 (describing how earlier NIH investment proved crucial to development of new vaccines)
[60] Because publicly available information is extremely limited, these are rough estimates. In addition, since many steps are being conducted simultaneously in the pandemic response, these timelines also include the time for large-scale process development. Once corporations have identified the most efficient way to conduct large-scale manufacturing, this could further reduce timelines for transfer—though these processes may need to be adapted depending on the recipient.
[61] Emergent BioSolutions Signs Agreement to be U.S. Manufacturing Partner for AstraZeneca’s COVID-19 Vaccine Candidate, Emergent BioSolutions (Jun. 11, 2020), https://tinyurl.com/y4bevhpz
[62] Stephanie Baker and Cynthia Koons, Inside Operation Warp Speed’s $18 Billion Sprint for a Vaccine, Bloomberg (Oct. 29, 2020), https://tinyurl.com/y62uhnoz
[63] An Indian firm starts mass-producing an unproven covid-19 vaccine, The Economist (Apr. 28, 2020), https://tinyurl.com/ya3vypqx Serum had also made earlier informal agreement with Oxford in April, but there is no publicly available information suggesting that technology transfer had begun that early.
[64] Serum to apply for licence to manufacture Oxford vaccine in a week, says CEO Adar Poonawalla, Press Trust of India (Jul. 21, 2020), https://tinyurl.com/y6rnkr6b
[65] Produced 40 Million Doses Of AstraZeneca Covid Vaccine: Serum Institute, All India/Reuters (Nov. 12, 2020), https://tinyurl.com/y6t3t4qn
[66] Moderna and Lonza Announce Worldwide Strategic Collaboration to Manufacture Moderna’s Vaccine (mRNA-1273) Against Novel Coronavirus, Moderna (May 1, 2020), https://tinyurl.com/y36l4rqy and 2020 Moderna SEC 2nd Quarter 10-Q, Moderna (Jun. 30, 2020), https://tinyurl.com/yyy9zc4b
[67] John Miller, Lonza confident of 2020 target for Moderna COVID-19 vaccine supply, Reuters (Sep. 29, 2020), https://tinyurl.com/yxqe2gxu
[68] Cormac O’Sullivan, Paul Rutten, and Caspar Schatz, Why tech transfer may be critical to beating COVID-19, McKinsey (Jul. 23, 2020), https://tinyurl.com/y3j57h5x
[69] MSF welcomes Fiocruz’s step towards transparency of AstraZeneca deal (Nov. 3 2020), https://msfaccess.org/msf-welcomes-fiocruzs-step-towards-transparency-astrazeneca-deal
[70] Similar results were obtained for manufacturing drug product (i.e., the finished dosage including container).
[71] Richard Hatchett, COVID vaccines development & manufacturing, CEPI/GAVI/WHO (Nov. 4, 2020), https://tinyurl.com/y4jxmz3m
[72] Some with COVAX, some between manufacturers facilitated by COVAX. Some groups have signed more than one deal, partially inflating participation.
[73] Pagliusi, Sonia, et al, Emerging Manufacturers engagements in the COVID-19 vaccine research, development and supply, Vaccine (Jul. 22, 2020), https://tinyurl.com/y6yg5sgg. (19/37.) Some DCVMN members not participating in the COVID response reported very limited capacity. Some developing country manufacturers are also not members of DCVMN.
[74] Global Partnerships and International Preparedness: Challenges and Innovative Partnering, ASPR (Oct. 2016), https://tinyurl.com/y23ub6y3
[75] Cormac O’Sullivan, Paul Rutten, and Caspar Schatz, Why tech transfer may be critical to beating COVID-19, McKinsey (Jul. 23, 2020), https://tinyurl.com/y3j57h5x
[76] COVID-19 technology access pool, WHO, https://tinyurl.com/ybavhba6
[77] Price, W. Nicholson, Arti K. Rai, and Timo Minssen, Knowledge transfer for large-scale vaccine manufacturing, Science 369.6506 (Aug. 21, 2020): 912-914. (Chemistry, manufacturing and controls.)
[78] Different but related statutory regimes apply depending on whether the federal government helped fund the development of an invention, or whether federal scientists helped develop the invention.
[79] Zain Rizvi, BARDA Funding Tracker, Public Citizen (Nov. 2, 2020), https://www.citizen.org/article/barda-funding-tracker/
[80] Kathryn Ardizzone, $1.8 Billion Sanofi Vaccine Contract Contains International Reference Pricing Clause, Preserves Standard IP and Data Rights, Knowledge Ecology International (Dec. 2, 2020), https://www.keionline.org/34776
[81] Hillary Chen, US Government First to Share Patents with Medicines Patent Pool, The White House (Sep. 30, 2010), https://tinyurl.com/yxsalos9
[82] Since patent laws are national, Section 1498 would authorize domestic use. But it would send an important signal for other countries to use similar authorities. For a list of relevant patents, see e.g., Mario Gaviria and Burcu Kilic, BioNTech and Pfizer’s BNT162 Vaccine Patent Landscape (Nov. 16, 2020), https://www.citizen.org/article/biontech-and-pfizers-bnt162-vaccine-patent-landscape/ and mRNA-1273 Vaccine Patent Landscape (For NIH-Moderna Vaccine), https://www.citizen.org/article/modernas-mrna-1273-vaccine-patent-landscape/.
[83] For example, 35 USC 202, 35 USC 209, 15 USC 3710a.
[84] Some agencies have said that the Bayh-Dole Act does not apply to certain kinds of Other Transaction Authority agreements.
[85] A Prescription for Excessive Drug Pricing: Leveraging Government Patent Use for Health, Yale Journal of Law and Technology (2017), https://tinyurl.com/y4aobrue
[86] Testimony of Dr. Julie L. Gerberding, U.S. House of Representatives (Jul. 21, 2020), https://tinyurl.com/y6xurl8s
[87] Id.
[88] Richard Hatchett, COVID vaccines development & manufacturing, CEPI/GAVI/WHO (Nov. 4, 2020), https://tinyurl.com/y4jxmz3m
[89] Id.
[90] Alex Azar, HHS Secretary Azar: Why Operation Warp Speed is a Made-in-America story, Fox Business (Oct. 22, 2020), https://tinyurl.com/y2onq58x
[91] Moncef Slaoui and Matthew Hepburn, Developing safe and effective Covid vaccines—Operation Warp Speed’s strategy and approach, New England Journal of Medicine 383.18 (Aug. 26, 2020, https://www.nejm.org/doi/full/10.1056/NEJMp2027405
[92] Pierre-Yves Oehrli, Lonza To Start Manufacturing Moderna’s COVID-19 Vaccine This December, Switzerand Global Enterprise (Oct. 7, 2020), https://tinyurl.com/y44atn7b
[93] FACT SHEET: Update on the Ebola Response, The White House (Dec. 2, 2014), https://tinyurl.com/y56reprg
[94] The Defense Production Act of 1950, FEMA, https://tinyurl.com/y5ydrq4c
[95] Stephanie Baker and Cynthia Koons, Inside Operation Warp Speed’s $18 Billion Sprint for a Vaccine, Bloomberg (Oct. 29, 2020), https://tinyurl.com/y62uhnoz
[96] 42 U.S.C § 247d–7e (“The Secretary may assess the feasibility and appropriateness of establishing, through contract, grant, cooperative agreement, or other transaction, an arrangement with an existing research center in order to achieve the goals of this section. If such an agreement is not feasible and appropriate, the Secretary may establish one or more federally-funded research and development centers, or university-affiliated research centers, in accordance with section 3304(a)(3) of title 41.”). Advanced research and development is defined as “related to manufacturing the product on a commercial scale.”
[97] HHS’s Center for Innovation in Advanced Development and Manufacturing, Medical Countermeasures, https://tinyurl.com/y54q4jka
[98] President George W. Bush’s 2003 State of the Union Address, The White House (Jan. 28, 2003), https://tinyurl.com/y4kkqw5d
[99] Results and Impact — PEPFAR, U.S. Department of State, https://www.state.gov/results-and-funding-pepfar/